没有合适的资源?快使用搜索试试~ 我知道了~
Water-medium Barbier Reactions over the Mesoporous Pd(II) Organo...
需积分: 0 0 下载量 43 浏览量
2020-02-19
14:35:51
上传
评论
收藏 524KB PDF 举报
温馨提示

试读
11页
高稳定类结晶壁介孔Pd(II)有机金属催化水介质Barbier反应的研究,何文翰,张昉,采用表面活性剂自组装合成联苯基和Pd(II)有机金属的PMO类型催化剂。该催化剂具有大比表面积、介孔结构以及强的表面疏水性,在水介质
资源推荐
资源详情
资源评论

http://www.paper.edu.cn
- 1 -
中国科技论文在线
Water-medium Barbier Reactions over the Mesoporous
Pd(II) Organometal Catalyst with Crystal-like Pore Walls
and High Stability
He Wenhan, Zhang Fang, Li Hexing, Huang Jianlin
(Department of Chemistry, Shanghai Normal University, ShangHai 200234)
Foundations: 教育部(博士点基金)(No.20070270001)
Brief author introduction:何文翰(1984-),男,环境催化
Correspondance author: 李和兴(1963-),男,教授,环境催化. E-mail: Hexing-L[email protected].cn
Abstract: A new approach was developed to prepare an immobilized Pd(II) organometallic catalyst
with mesoporous structure by surfactant-directed co-condensation between biphenyl-bridged
organosilane and Pd(II) organometallicsilane. Both the Pd(II) organometallic complex and the biphenyl
groups were embedded in the pore wall, which could diminish the blockage of mesoporous channels.
As a result, such catalyst exhibited high activity and selectivity in water-medium Barbier reactions
owing to the high surface area, large pore size and strong surface hydrophobicity resulting from
biphenyl functionaliztion, which facilitated the diffusion and adsorption of organic reactant molecules
on the active sites. Meanwhile, the as-prepared catalyst also displayed long durability owing to the
crystal-like mesoporous wall resulting from the π-π stacking of biphenyl groups, which could protect
the mesoporous structure from collapsing in aqueous Barbier reactions.
Keywords:Environmentalcatalysis;biphenylene-bridgedPMOmaterials;crystal-likeporewalls;mesoporo
us Pd(II) organometallic catalyst;water-medium Barbier reactions
0 Introduction
Environmental concerns associated with chemical processes have encouraged the
development more environmentally friendly (greener) organic reactions. In recent years,
reactions that take place in the environmentally clean, safe and cheap solvent water have received
considerable interest
[1]
. However, formidable challenges remain to be addressed as results of the
solubility and diffusion limits of organic substances in aqueous solution.
Homogenous catalytic processes, which in general occur in high yield and under mild
reaction conditions, are dominant strategies in chemical catalysis. Nevertheless, the industrial
use of homogeneous catalysts is relatively limited because of difficulties in separation and reuse
that eventually adds cost and cause environmental pollution from heavy metallic ions
[2]
. A great
number of attempts have been made to prepare immobilized heterogeneous catalysts which show
in some cases superiority in separation and repetitive uses
[3]
. However, such heterogeneous
catalysts cannot match in activity and selectivity with their corresponding homogeneous
counterparts due to the low dispersion degree of active sites. Mesoporous materials with large
surface area, ordered pore channels, and highly dispersed active sites afford powerful supports for
anchoring homogeneous catalysts. Previously, we reported the preparation of metal and
organometallic catalysts anchored on the SBA-15 and FDU-12 supports and their applications in
water-medium organic reactions
[4]
. We found that functionalization of silica supports with
organic groups could greatly promote the adsorption of organic reactant molecules on the catalysts
owing to the enhanced surface hydrophobicity, leading to high activity and selectivity. However,
the traditional immobilization of homogeneous catalysts by coordinating the metallic ions with
organic ligands incorporated into the silica support usually blocks the pore entrance and pore
channels. Furthermore, functionalization of silica support with organic groups inevitably blocks
the pore channels and causes damage to pore structure. Accordingly, the catalytic efficiency

http://www.paper.edu.cn
- 2 -
中国科技论文在线
decreases dramatically due to the diffusion limit. Recently, we incorporated a Pd(II)
organometallic catalyst into the periodic mesoporous organosilicas (PMO)
[5]
, which effectively
diminishes the blockage of pore channels since the organic groups are uniformly embedded into
the silica wall. More recently, we have developed a new approach to prepare immobilized Ru(II)
catalyst with mesoporous structure via co-condensation of Ru(II) organometallicsilane with TEOS
in the presence of a surfactant self-assembly
[6]
. Although these materials possessed high activity
and selectivity, owing to poor hydrothermal stability these catalysts usually exhibited short
durability in water-medium organic reactions.
It has been demonstrated that the PMO materials with crystal-like walls exhibit stronger
hydrothermal stability than those with amorphous silica walls
[7]
. Up to now, nearly all PMO
materials with crystalline mesoporous walls are prepared under basic condition
[7, 8]
. This is
unfavorable for preparing immobilized organometallic catalysts via co-condensation due to the
decomposition of organometallicsilane under basic conditions. In this paper, we wish to disclose a
new Pd(II) organometallic catalyst immobilized in a biphenylene-bridged PMO support prepared
by co-condensation under acidic conditions. This catalyst exhibited mesoporous structure with
crystal-like walls, having both the high activity and the strong durability during water-medium
Barbier reactions.
1 Experimental
1.1 Catalyst preparation
Firstly, a Pd(II) organometallicsilane was synthesized in dry and oxygen-free argon
atmosphere using Schlenk techniques. In a typical run of synthesis, 2.7 g PPh
2
CH
2
CH
2
Si(OEt)
3
was added slowly into 25 ml anhydrous toluene containing 1.0 g PdCl
2
(COD). After being stirred
for 24 h at 25
o
C, the toluene was evaporated to about 5.0 ml. A yellow solid product was then
obtained by adding hexane. According to ICP analysis and 1H NMR (CD
2
Cl
2
) analysis: 7.2-7.5,
7.6-7.8 (3 m, 20 H, ArH), 3.7 (m, 12 H, OCH
2
), 2.5 (m, 4H, CH
2
P), 1.1 (m, 18 H, CH
3
), 0.79 (m, 4
H, SiCH
2
) the composition of the Pd(II) organometallicsilane was determined as
PdCl
2
[PPh
2
(CH
2
)
2
Si(EtO)
3
]
2
.
Then, the immobilized Pd(II) organometallic catalyst was synthesized through
surfactant-directed co-condensation between the 4, 4’-bis(triethoxysilyl)-biphenyl (BTESBP) and
the as-prepared PdCl
2
[PPh
2
(CH
2
)
2
Si(EtO)
3
]
2
under acidic solution. Briefly, certain mount of
BTESBP was added dropwise into 20 ml aqueous solution containing 0.50 g P123 (EO
20
PO
70
EO
20
)
and 0.20 M HCl. After pre-hydrolysis for 2.5 h at 40
o
C, 4.0 ml THF solution containing required
amount of PdCl
2
[PPh
2
(CH
2
)
2
Si(EtO)
3
]
2
was added into the mixture. The initial molar ratio in this
system is fixed at Si : P123: HCl : H
2
O = 1 : 0.017 : 0.80 : 218, where Si refers to the total silica
source including both the BTESBP and the PdCl
2
[PPh
2
(CH
2
)
2
Si(EtO)
3
]
2
. The mixture was stirred
for 24 h at 40
o
C and transferred to a sealed autoclave. After hydrothermal treatment at 100
o
C for
another 24 h, the yellow precipitate was filtrated and washed thoroughly with water, following by
vacuum drying at 80
o
C overnight. The surfactant, together with both the Pd(II) species and
organic residues physisorbed on the solid were then removed by refluxing in 0.5mol/L
HCl/ethanol at 80
o
C for 24 h. The as-prepared sample was denoted as Pd-PPh
2
-PMO(Ph-Ph)-X,
where X refers to the different Pd loadings which could be adjusted by changing the amount of
PdCl
2
[PPh
2
(CH
2
)
2
Si(EtO)
3
]
2
in the mother mixture. As a reference, Pd-PPh
2
-SBA-15(Ph) was
synthesized in the same way except for the replacement of BTESBP by the mixture of TEOS and
phenyltrimethoxysilane (PTMS) which was co-condensed with palladium saline in the acid
http://www.paper.edu.cn
- 3 -
中国科技论文在线
condition, meanwhile with the same procedure in mind, the Pd-PPh
2
-PMO(Et) was synthesized by
using bis(triethoxysilyl)ethane (BTESE) to replace the BTESBP as the precursor in the acid
condition.
1.2 Characterization
The Pd(II) loadings were determined by an inductively coupled plasma optical emission
spectrometer (ICP, Varian VISTA-MPX). Solid state nuclear magnetic resonnce (NMR) spectra
were recorded on a Bruker AV-400 spectrometer at 79.5, 100.6 and 169.3 MHz, respectively. The
X-ray powder diffraction (XRD) patterns were obtained on a Rigaku D/Max-RB diffractometer
with CuKα radiation. Transmission electron microscopy (TEM) morphologies were observed on a
JEOL JEM2010. N
2
adsorption–desorption isotherms were measured at -196
o
C on a
Quantachrome NOVA 4000e analyzer after being degassed at 100
o
C overnight. Based on
adsorption-branches, the specific surface area (S
BET
), pore size distribution, pore volume (V
P
) and
average pore diameter (D
P
) were calculated by using the multiple-point Brunauer–Emmett–Teller
(BET) and the Barrett–Joyner–Halenda (BJH) models, respectively. The X-ray Photoelectron
Spectra (XPS) were analyzed on the PHI 5000 VersaProbe. All the binding energy values were
calibrated by using C
1S
= 284.8 eV as a reference.
1.3 Activity test
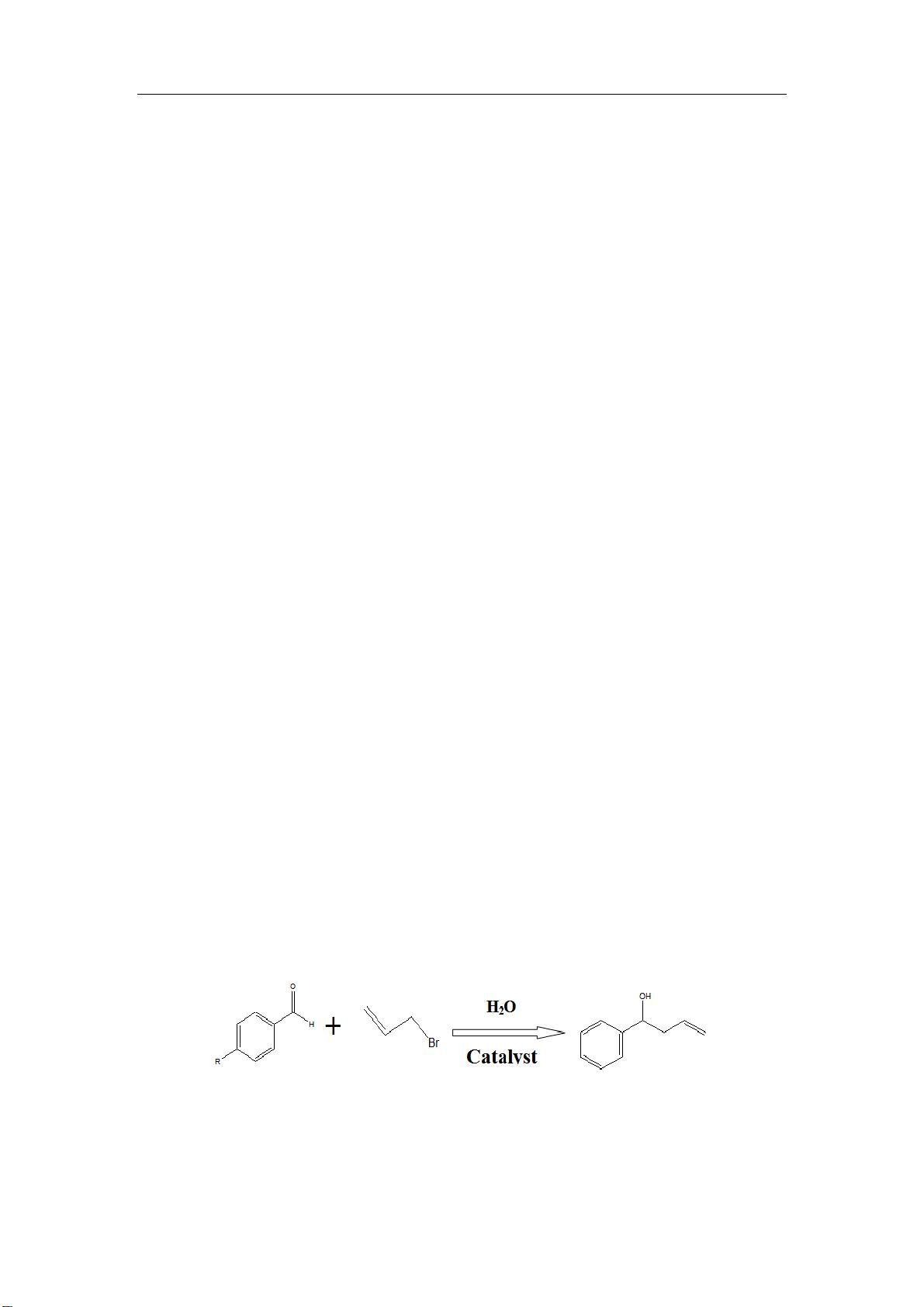
The water-medium Barbier reaction (see Scheme 1) was carried out at 50
o
C in a 10 ml flask
containing 25 ul benzaldehyde, 150 ul allyl bromide, 0.45 g SnCl
2
and 5.0 ml H
2
O, and a catalyst
with 0.065mmol Pd(II). The organic substances were protected from evaporation by refluxing.
After reaction under vigorous stirring for 12 h, the reaction mixture was extracted by toluene and
dried by anhydro-magnesium sulfate. The liquid products were quantitatively analyzed on a gas
chromatograph (GC, Agilent 1790) equipped with a FID and a JWDB-5 95% dimethyl
1-(5%)-diphenylpolysiloxane column at 373 K in N
2
flow, from which both the conversion,
selectivity and yield to target product were determined by using internal standards. The conversion
was calculated based on benzaldehyde since allyl bromide was greatly excess. The reproducibility
was checked by repeating each result at least three times and was found to be within acceptable
limits (±5%).
In order to determine the catalyst durability, the target catalyst was allowed to settle down
after each run of reactions and the clear supernatant liquid was decanted slowly. After being
washed with water, acetone and ethyl ether for three times each, followed by vacuum drying at
80
o
C overnight, the catalyst was re-used with fresh charge of solvent and reactant for subsequent
reaction under the same conditions. The content of Pd species both in the solution and on the solid
support were determined by ICP analysis to show the leach of Pd(II) active sites during each run
of reactions.
Sch. 1 The formula of Barbier reaction equation
2 Result and Discussion
The Pd(II) loadings in different catalytic reactions were determined by ICP analysis and
剩余10页未读,继续阅读
资源评论

weixin_38687928
- 粉丝: 2
- 资源: 950
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功
