没有合适的资源?快使用搜索试试~ 我知道了~
温馨提示

试读
17页
背景:视黄醇(RE)深入参与皮肤加工,因此在化妆品中被广泛配制,主要是作为抗衰老成分。 尽管具有疗效,但RE的安全性仍存在争议。 目标:视黄醇(PRE)复合物是由两种RE的前体β-胡萝卜素和烟酰胺配制而成的,其目的是提供类似视黄醇的皮肤有益健康的特性,前提是假定皮肤酶能够当场安全地供应RE。 方法:在暴露于PRE和RE之前和之后,使用各种皮肤模型测量透明质酸,肿瘤坏死因子α(TNFα)和白细胞介素1α(IL-1α)的表达水平。 进行了全基因组微阵列,并分析了受影响的基因和途径。 结果:成纤维细胞暴露于PRE后,透明质酸的天然合成显着提高。 通过细胞因子在离体皮肤上的表达证明了皮肤安全性,RE应用可导致TNFα和IL-1α升高。 相反,PRE可显着降低TNFα,而IL-1α不受影响。 这些结果确立了PRE与RE的皮肤安全优势。 在皮肤等效物上检查的微阵列结果表明,PRE参与了炎症减轻。 结论:部分支持将可再生能源前体制成可再生能源的安全来源。 PRE具有与RE平行的皮肤益处,而PRE的特性则建议对皮肤更安全。
资源推荐
资源详情
资源评论

Journal of Cosmetics, Dermatological Sciences and Applications, 2020, 10, 59-75
https://www.scirp.org/journal/jcdsa
ISSN Online: 2161-4512
ISSN Print: 2161-4105
DOI:
10.4236/jcdsa.2020.102007 Jun. 1, 2020 59 J. Cosmetics, Dermatological Sciences and Applications
Safe Retinol-Like Skin Biological Effect by
a New Complex, Enriched with
Retinol Precursors
Ze’evi Ma’or
1,2
, Dror Cohen
1,2
, Ya’ara La’or-Costa
1,2
, Meital Portugal-Cohen
1,2
1
Ahava Dead Sea Laboratories, Lod, Israel
2
The Skin Research Institute, the Dead-Sea and Arava Science Center, Masada, Israel
Abstract
Background:
Retinol (RE) is deeply involved in skin processes, therefore it is
widely formulated in cosmetics, primarily as an anti-aging in
gredient. Despite
its efficacy, the safety profile of RE is controversial.
Objectives:
Pretinol (PRE)
complex was formulated with two RE precursors,
β
-Carotene and Niacinamide,
in order to deliver retinol-like skin benefits with healthier characteristics, as-
suming that skin enzymes will enable safe RE supply on spot.
Methods:
The ex-
pression levels of hyaluronic acid, Tumor Necrosis Factor alpha (TNF
α
) and In-
terleukin 1 alpha (IL-1
α
), were measured using various skin models before and
after exposure to PRE and RE. Full genome microarray was performed and the
affected genes and pathways were analyzed.
Results:
Following fibroblasts ex-
posure to PRE, the natural synthesis of hyaluronic acid is significantly elevated.
Skin safety, demonstrated via cytokines expression on
ex-vivo
skin, results with
TNF
α
and IL-1
α
elevation by RE application. In contrary PRE significantly re-
duces TNF
α
while IL-1
α
is not affected. These results establish skin safety ad-
vantage of PRE
vs
RE. Microarray results examined on skin equivalents reveal
the involvement of PRE in inflammatory attenuation.
Conclusions:
Formulat-
ing RE precursors as a safe source for RE is partially supported. PRE presents
a skin benefit in parallel to RE, while PRE
characteristics are suggested to be
safer to skin.
Keywords
Retinol, Hyaluronic Acid,
β
-Carotene, Skin, Inflammaging, Irritation
1. Introduction
Vitamin A, the most multifunctional vitamin in the human body, constitutes a
How to cite this paper:
Ma’or, Z.,
Cohen,
D
., La’or-Costa, Y. and Portugal-Cohen, M.
(20
20) Safe Retinol-Like Skin Biological Effect
by a New Complex, Enriched with Retinol
Precursors
.
Journal of Cosmetics
,
Dermat
o-
logical
Sciences and Applications
,
10
, 59-75.
https://doi.org/10.4236/jcdsa.2020.102007
Received:
April 19, 2020
Accepted:
May 28, 2020
Published:
June 1, 2020
Copyright © 20
20 by author(s) and
Scientific
Research Publishing Inc.
This work is licensed under the Creative
Commons Attribution International
License (CC BY
4.0).
http://creativecommons.org/licenses/by/4.0/
Open Access

Z. Ma’or et al.
DOI:
10.4236/jcdsa.2020.102007 60 J. Cosmetics, Dermatological Sciences and Applications
group of organic lipid-soluble compounds comprising retinol (RE) and its de-
rivatives, mainly the RE esters, retinyl palmitate and retinyl acetate [1] [2] [3].
RE is deeply involved in growth and maintenance due to its cellular contribution
to cell proliferation and differentiation from early embryogenesis to adulthood
[4]. RE participates in many skin biological processes in epidermis and dermis
and hence is formulated in a lot of cosmetic preparations, mainly as anti-aging
and skin brightening agent [5] [6]. Its contribution to alleviating acne symp-
toms was proven and as a result it is widely formulated in dermo-cosmetic prod-
ucts aiming to treat acne-prone and oily to problematic skin conditions [7] [8].
Despite its proven efficacy, the safety assessment of RE is still controversial [9].
According to some reports, the topical applications of RE containing skin prep-
arations might trigger severe side effects, such as dryness and irritation, skin
color changes and sensitivity to sunlight [10]. This fragile safety status affects RE
acceptance in cosmetics. Chemists all over the world are searching for new ways
to develop safe RE cosmetics,
i.e.
formulating skin preparations with proven RE
skin benefits, that are free off RE related side effects. Sophisticated delivery sys-
tems for RE, such as liposomes, nano and micro encapsulations were proposed
and clinically tested [11], but these technologies did not solve the fragile status
related to RE containing formulae. In some cases, the situation was even wor-
sened, especially if enhanced RE percutaneous penetration was measured [12].
In order to achieve a desired safe retinol-like skin activity, the idea to formulate
its precursors was raised, assuming that the natural skin enzymatic activity will
effectively convert these precursors, and will enable the skin tissue a safe level of
RE supply “on the spot”.
β
-Carotene was chosen to serve as a successful RE pre-
cursor and a bio-available RE source [13], as it is a safe compound, that when
processed by epidermal enzymes, is converted into retinyl esters to supply skin
with RE [14]. Combined with Niacinamide, known as Niacin and Vitamin B3, as
it is also involved in the final stage of the skin’s natural process of retinol biosyn-
thesis, mediated by RE dehydrogenase enzyme with Nicotinamide adenine di-
nucleotide Coenzyme A (NAD Co-A) [15] [16]. The natural skin process of RE
biosynthesis from
β
-Carotene and Niacinamide as precursors is described in
Figure 1 below.
A new complex named “pRETINOL
TM
” (PRE) was designed containing
β
-
carotene and Niacinamide as RE precursors, with the addition of two Dead Sea
natural actives, Dead Sea mineral rich water and an extract of
Dunaliella Salina
,
Dead Sea alga. Dead Sea water skin benefits are well established and are widely
formulated in cosmetics, mainly as a moisturizing humectant and an anti-aging
active ingredient [17] [18].
Dunaliella Salina
, a key micro-flora microorganism
of The Dead Sea, was first discovered by Volcani in the Dead Sea saturated saline
water in 1940 [19]. Following
Dunaliella Salina
exposure to sun the alga accu-
mulates large quantities of
β
-carotene at its cell periphery [20]. The
β
-carotene
accumulated level depends on water salinity, temperature and light intensity and
could accomplish up to 10% of alga dry weight [19].
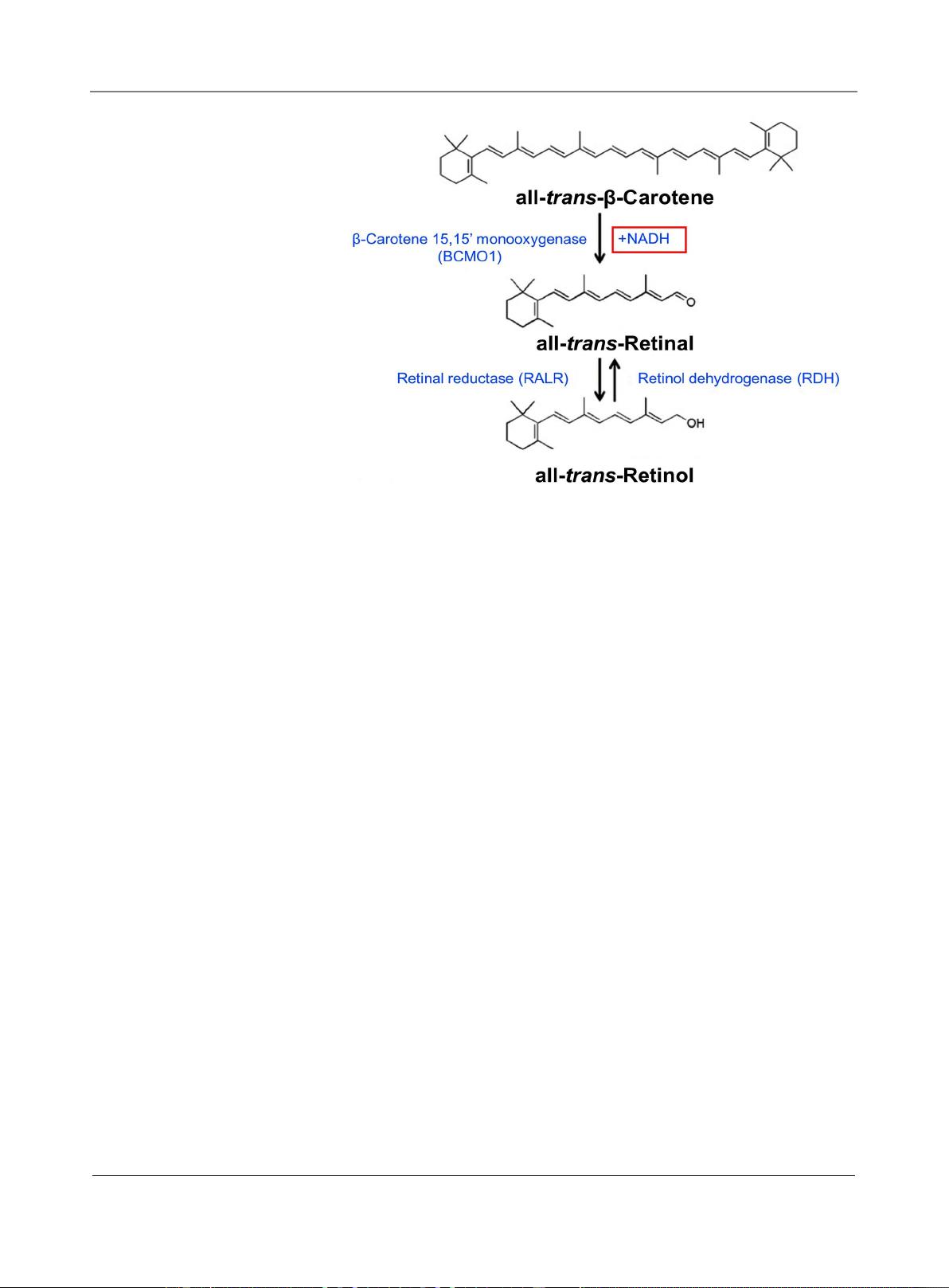
Z. Ma’or et al.
DOI:
10.4236/jcdsa.2020.102007 61 J. Cosmetics, Dermatological Sciences and Applications
Figure 1. Retinol biosynthesis [2].
The
Dunaliella Salina
natural synthesis of
β
-carotene is believed to assist survival
in the extreme Dead Sea conditions, especially when the exposure to harmful sun
irradiation is high [21]. The bioactivities and skin safety of PRE complex, com-
prised of Dead Sea water,
Dunaliella Salina
alga, and RE precursors:
β
-carotene
and Niacinamide, were elucidated. The expression levels of relevant biomarkers
were measured using various skin models:
In vitro
Human dermal fibroblasts
cell cultures, reconstructed 3D skin equivalent and
ex-vivo
human skin organ
culture. In this work we present our test results and the new complex capabilities
to introduce safe RE cosmetics.
2. Methods
2.1. Preparation of Test Materials
2.1.1. pRETINOL—A Complex Enriched with Retinol Precursors
pRETINOL (PRE) was prepared as a complex of 4 different active materials added
and mixed together via vortex, including concentrated
β
-Carotene (
β
-Carotene
10% CWS/S dispersed in a starch coated matrix. DSM), Niacinamide/pyridine-
3-carboxamide (KOYOWA HAKKO Europe GmbH),
Dunaliella Salina
commer-
cial aqueous extract (PHEPA
-CTIVE, DSM), and Dead Sea water extract (Os-
moter
TM
by AHAVA—Dead Sea Laboratories Ltd.) collected from the Dead Sea
lake as describe at Portugal-Cohen
et al.
[17]. The relative ration of four PRE com-
plex compounds is 0.0156
β
-Carotene: 1 Niacinamide: 30
Dunaliella Salina
aqueous
extract: and 5 Dead Sea water extract (w/w/v/v). PRE was diluted before applica-
tion with distilled water according to test protocols. For HSOC skin test, PRE
complex was diluted with distilled water before skin application to a final con-
centration of 3.6% w/w. On fibroblasts cell culture test, PRE complex was added
to fibroblast cell culture media to a final concentration of 1.44% (v/v).

Z. Ma’or et al.
DOI:
10.4236/jcdsa.2020.102007 62 J. Cosmetics, Dermatological Sciences and Applications
2.1.2. Retinol
Retinol (RE) (BASF, Retinol 50C), contains 46.6% all-trans retinol, 49% poly-
sorbate 20, 1.0% of Butylhydroxyanisole, and 3.4% of Butylhydroxytoluene. RE
was diluted with ethanol according to test protocols. On HSOC skin test, 3 µl of
1% (w/w) RE in ethanol, was topically applied. On dermal Fibroblasts test, 1.0
µM of RE were added to fibroblast culture medium.
2.2. Ex-Vivo Human Skin Organ Culture Preparation Protocols
Ex-vivo
human skin organ culture (HSOC) was prepared as described in Por-
tugal
et al.
[22]. Human skin cultures were obtained from healthy females, aged
37 - 60, undergoing abdominal plastic surgery in accordance with Hospital Eth-
ics Committee approval. 0.64 cm
2
fixed-size of explant skin pieces were cut from
donated skin tissue, placed with dermal side down in culture medium Dulbec-
co’s Modified Eagle’s High D glucose medium (DMEM), supplemented with 100
U/ml penicillin and 100 μg/ml streptomycin), and incubated overnight at 37˚C
with 5% CO
2
for 24 h of recovery. Following incubation
ex-vivo
HSOC pieces
were treated topically with tested materials.
2.3. Human Primary Fibroblast Cell Culture Protocols
In-vitro
human primary dermal fibroblast cell culture was obtained based on the
protocol described by Limat
et al.
[23]. Human primary fibroblast cells were
grown in cultures to 80% confluency with DMEM culture medium, enriched
with 10% fetal bovine serum 100 U/ml penicillin, and 100 U/ml streptomycin.
The cells were cultivated in 6 wells plates. Tested materials were diluted before
application as follows: PRE complex was diluted × 2.5 with culture medium
DMEM. RE was added to the culture medium at a final concentration of 1.0 µM.
Fibroblast cells were incubated at 37˚C, 5% CO
2
for 48 h. After incubation, the
cells were extracted using RIPA Lysis Buffer System (Santa Cruz Biotechnology)
collected to a blending Eppendorf and ground using stain steal beads and a bul-
let blender tissue homogenizer (Bullet blender blue, Next Advance) at maximum
speed for 5 min. The samples were centrifuged at 4˚C for 15 min at 5000 rpm,
and the supernatant was collected. Protein content on epidermis lysate was de-
termined using BCA Protein assay [24]. The protein level for all samples was di-
luted with PBS to reach a final concentration of 0.1 ng/ml. Samples were tested
for Hyaluronic Acid (HA) using ELISA assay kit (R&D Systems). Results are pre-
sented as ng/ml of HA per 1.0 µg of protein.
2.4. Reconstructed Skin Model
Full Thickness reconstructed skin tissues (MatTek EFT-400 full-thickness skin
tissues) was used as described in Portugal
et al.
[25]. Skin pieces were placed into
a 6-well plate containing 2.5 ml of assay medium and incubated overnight at 37˚C
and 5% CO
2
. Then, the assay medium was replaced with 5 ml of fresh medium
and the tissues were treated topically with tested materials for 48 h. At the end of
剩余16页未读,继续阅读
资源评论

weixin_38678394
- 粉丝: 2
- 资源: 861
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功
