没有合适的资源?快使用搜索试试~ 我知道了~
巴克莱-美股-医疗保健行业-美国生命科学与诊断:巴克莱医疗会议上关于工具与诊断的十大争论-35-45页.pdf
需积分: 0 0 下载量 119 浏览量
2023-07-26
11:49:07
上传
评论
收藏 940KB PDF 举报
温馨提示

试读
45页
巴克莱-美股-医疗保健行业-美国生命科学与诊断:巴克莱医疗会议上关于工具与诊断的十大争论-35-45页.pdf
资源推荐
资源详情
资源评论

Equity Research
5 March 2019
CORE
Barclays Capital Inc. and/or one of its affiliates does and seeks to do business with
companies covered in its research reports. As a result, investors should be aware that the
firm may have a conflict of interest that could affect the objectivity of this report. Investors
should consider this report as only a single factor in making their investment decision.
PLEASE SEE ANALYST CERTIFICATION(S) AND IMPORTANT DISCLOSURES BEGINNING ON PAGE 37.
Restricted
- Internal
U.S. Life Science Tools & Diagnostics
Top 10 Tools & Dx Debates for the
Barclays Healthcare Conference
Next week, we are hosting the
Barclays Global Healthcare Conference
in Miami
Beach, FL. We’re expecting healthy investor debates amongst our participating
companies, including Life Science Tools: BRKR, ILMN, MTD, PKI, RGEN, TECH, & TMO;
Diagnostics: EXAS, HOLX, LMNX, OXFD, QDEL, & QGEN; and Labs/CROs/CDMOs:
CTLT, CRL, DGX, ICLR, IQV, & LH. The Top 10 Tools & Dx debates we see include:
1) How durable is the biopharmaceutical end market for Life Science Tools and the
CROs? We believe biopharma is a sustainable growth engine for the Life Science Tools
& CROs, driven by higher levels of new drug approvals and ample access to funding.
Additionally, we believe Thermo Fisher’s unique biopharma assets are well positioned
to benefit from the shift toward development of personalized therapies. Jack’s Job
Board: After two years of healthy new awards, we continue to see an acceleration in the
number of CRO employees being hired - with IQVIA standing out vs peers.
2) Are data strategies driving share shifts among CROs? As several CROs develop
“Next Generation” platforms for clinical development, the debate is around if and when
data offerings will transition from being a “nice to have” to a “need to have”. IQVIA’s
strong new business wins raise questions around whether the transition is approaching.
3) How durable is growth in the China end market? Strong regional growth supports
the argument that support for life science research is durable, though concerns persist.
4) What is driving contrasting diagnostic strength relative to the clinical lab
pressure? We’ve been perplexed at contrasting results between the labs and diagnostic
manufacturers. For Dx, the risk is reimbursement reductions bleed through over time.
5) Can the national labs cut enough costs to offset pricing pressure in 2019?
Significant rate headwinds increase focus on operational improvements to hit guidance.
6) When will clinical lab consolidation accelerate? We believe the confluence of lab
industry pressures should have an outsized impact on regional and hospital-based labs.
The key question is timing, following a slower year of deal-making in 2018.
7) Is the industry primed for increased M&A? 2018 was a slow year for acquisitions
by any measure. Looking to 2019, we believe there is a case to be made for increased
activity, given the fragmented landscape and clear opportunities for consolidation.
8) What is the next hot life science tool or diagnostic? We view an accelerated pace
of innovation as an investment positive for the entire group.
9) Which company is best positioned for a downturn? We believe our coverage is
more recession-proof than the broader market. The national labs screen as the most
defensive area, followed by the CROs, then Diagnostics and Life Science Tools.
10) Are Life Science Tools approaching peak positive sentiment? The group is now
trading at close to all-time highs. We believe the biggest fundamental risk is around the
Industrial end market – where macro datapoints have moderated.
Actions: We raise our TMO PT ($295, up from $280) and IQV ($160, up from $155).
INDUSTRY UPDATE
U.S. Life Science Tools & Diagnostics
NEUTRAL
Unchanged
For a full list of our ratings, price target and
earnings changes in this report, please see
table on page 2.
U.S. Life Science Tools & Diagnostics
Jack Meehan, CFA
+1 212 526 3909
jack.meehan@barclays.com
BCI, US
Mitchell Petersen
+1 212 526 3367
mitchell.petersen@barclays.com
BCI, US
Andrew Wald
+1 212 526 9436
andrew.wald@barclays.com
BCI, US
Barclays | U.S. Life Science Tools & Diagnostics
5 March 2019 2
Summary of our Ratings, Price Targets and Earnings Changes in this Report (all changes are shown in bold)
Company
Rating
Price
Price Target
EPS FY1 (E)
EPS FY2 (E)
Old
New
01-Mar-19
Old
New
%Chg
Old
New
%Chg
Old
New
%Chg
U.S. Life Science Tools & Diagnostics
Neu
Neu
IQVIA (IQV)
OW
OW
143.35
155.00
160.00
3
6.30
6.30
-
7.25
7.25
-
Thermo Fisher Scientific, Inc. (TMO)
OW
OW
264.06
280.00
295.00
5
12.15
12.15
-
13.75
13.75
-
Source: Barclays Research. Share prices and target prices are shown in the primary listing currency and EPS estimates are shown in the reporting currency.
FY1(E): Current fiscal year estimates by Barclays Research. FY2(E): Next fiscal year estimates by Barclays Research.
Stock Rating: OW: Overweight; EW: Equal Weight; UW: Underweight; RS: Rating Suspended
Industry View: Pos: Positive; Neu: Neutral; Neg: Negative

Barclays | U.S. Life Science Tools & Diagnostics
5 March 2019 3
CONTENTS
1) HOW DURABLE IS THE BIOPHARMACEUTICAL END MARKET FOR
LIFE SCIENCE TOOLS AND THE CROS? ....................................................... 4
Jack’s Job Board - CROs: Hiring Continues to Accelerate .................................................................... 7
2) ARE DATA STRATEGIES DRIVING SHARE SHIFTS AMONG CROS?11
3) HOW DURABLE IS GROWTH IN THE CHINA END MARKET? .......... 13
4) WHAT IS DRIVING CONTRASTING DIAGNOSTICS STRENGTH
RELATIVE TO CLINICAL LAB WEAKNESS? ................................................ 16
5) CAN THE NATIONAL LABS CUT ENOUGH COSTS TO OFFSET
PRICING PRESSURE IN 2019? ....................................................................... 17
6) WHEN WILL CLINICAL LAB CONSOLIDATION ACCELERATE? ....... 18
7) IS THE INDUSTRY PRIMED FOR INCREASED M&A? .......................... 21
8) WHAT IS THE NEXT HOT LIFE SCIENCE TOOL OR DIAGNOSTIC? 24
9) WHICH COMPANY IS BEST POSITIONED FOR A DOWNTURN? .... 25
10) ARE LIFE SCIENCE TOOLS APPROACHING PEAK POSITIVE
SENTIMENT? ..................................................................................................... 29
ACTIONS: RAISE PT ON THERMO FISHER TO $295, IQVIA TO $160 33
Barclays | U.S. Life Science Tools & Diagnostics
5 March 2019 4
1) HOW DURABLE IS THE BIOPHARMACEUTICAL END MARKET FOR LIFE SCIENCE
TOOLS AND THE CROS?
Given the importance of the biopharma end market for our coverage, the sustainability
of its growth remains a critical debate. While we believe investors often view this end
market as cyclical in nature, our view is that growth is more sustainable than the market
thinks. Amidst a more uncertain macro environment, we expect the biopharma end market
to remain resilient relative to other end markets, and to continue as a growth driver for our
coverage. There are a number of factors that improve our conviction into this dynamic:
1) breakthroughs in science are allowing for completely new classes of drugs to succeed, 2)
good science, combined with an accommodative FDA, are driving new drug approvals to
record levels, which should support future investment, 3) capital markets activity is creating
a robust funding environment, and 4) patent cliffs are driving investments by larger
biopharma companies as they aim to refresh drug pipelines.
Simply put, we believe the level of innovation that exists currently is unprecedented,
creating a sustainable growth engine for the Life Science Tools & CROs. Innovative new
drug mechanisms (i.e. cell and gene therapies) are enabling treatments that weren’t
possible previously. In an FDA statement from January 2019, the agency noted that it
expects to receive 200 cell and gene therapy INDs annually by 2020, and by 2025 it expects
10 to 20 approvals per year. Additionally, recent breakthroughs in genomic and
phonemic sciences, made possible by technological developments, have allowed
scientists to develop a more specialized view of individualized patient responses to
disease treatments. For example, advancements in next-generation sequencing (NGS)
have made it significantly more cost efficient to sequence the human genome. This is
enabling significant levels of data collection, leading researchers to gain a better
understanding of human disease. While these advancements lead to new R&D
breakthroughs, they also make R&D more efficient, as leveraging genetics to inform
selection of drug targets can improve success rates.
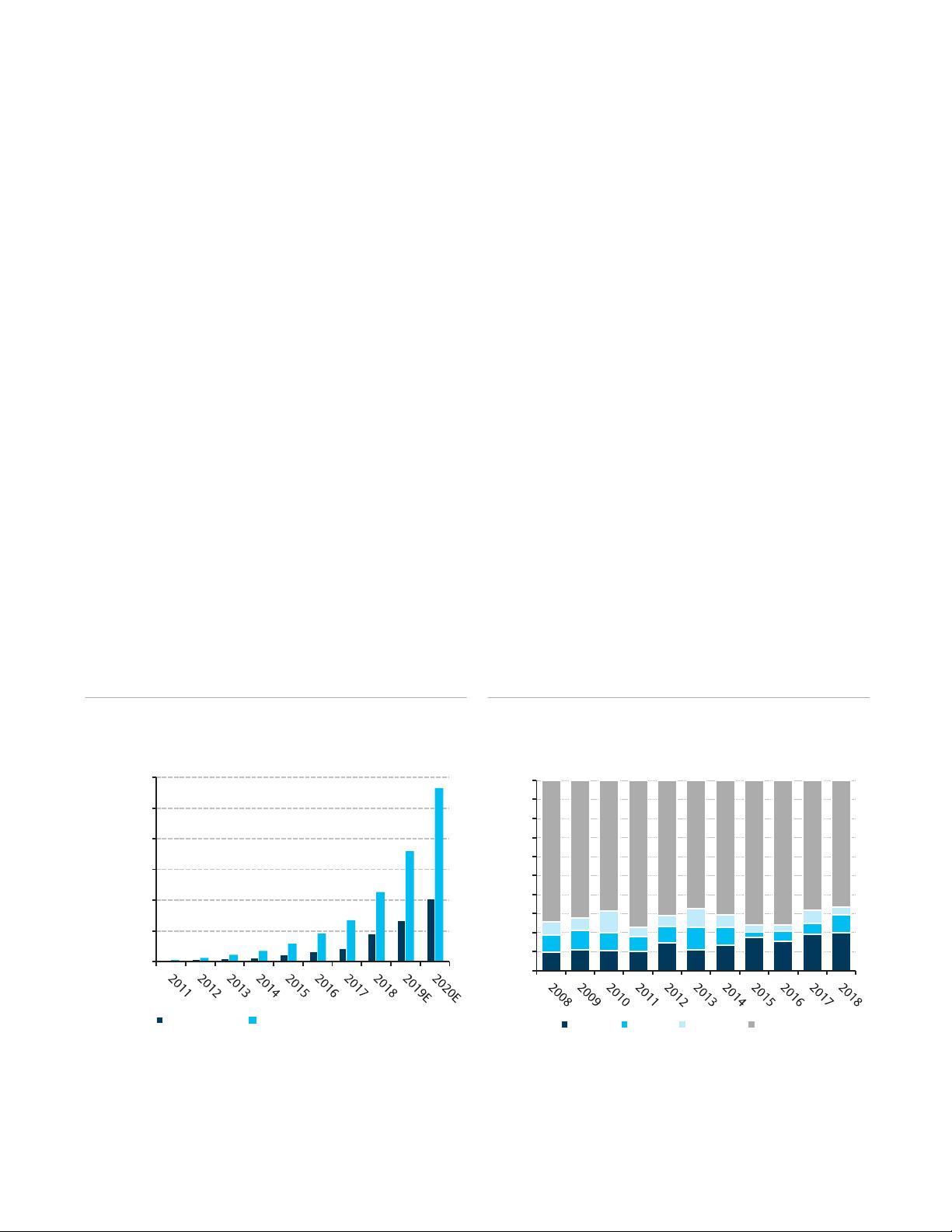
FIGURE 1
Barclays Estimates for Gigabases (Gb) Sequenced
FIGURE 2
Clinical Trial Patient Mix: Oncology Represents an Increasing
Portion of Trial Participants
Source: Company Documents, Barclays Research
Source: clinicaltrials.gov, Barclays Research
Data as of 12/31/2018
We believe higher levels of new drug approvals are supportive of future R&D
investment. Approved drugs create profits for the industry, which can ultimately be used to
drive sales (creating a manufacturing opportunity for the Life Science Tools) or invest in the
0
200,000
400,000
600,000
800,000
1,000,000
1,200,000
Gb Total (K)
Gb Cumulative (K)
10%
11%
10%
10%
14%
11%
14%
17%
16%
19%
20%
9%
10%
10%
8%
9%
12%
9%
3%
5%
6%
9%
7%
6%
11%
5%
6%
10%
6%
4%
3%
7%
4%
74%
72%
69%
77%
71%
67%
71%
76%
76%
68%
67%
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
Cancer
Cardio
Diabetes
Other
For the full FDA statement, see
here

Barclays | U.S. Life Science Tools & Diagnostics
5 March 2019 5
development of new therapies. We believe greater approval rates are due to a
combination of more successful (and targeted) therapies as well as a more
accommodative FDA. In 2018, 59 novel drugs were approved by the FDA (up 28% y/y),
representing a record for the industry.
FIGURE 3
CDER First Action Approval Rates
FIGURE 4
FDA Novel Drug Approvals Continue to Accelerate
Source: FDA
Note: As of 11/30/2018
Source: FDA, Barclays Research
Data as of 12/31/2018
Strong access to funding through the capital markets has spurred R&D investments by
SMiD biopharma companies. As described above, these companies are capitalizing on
scientific advancements allowing for the treatment of new, more targeted indications, often
with new mechanisms (i.e. cell or gene therapies). With improved funding, this class of
biopharma companies is helping to buoy end market demand. That said, given the
heightened volatility in the equity markets recently, it will be important to monitor the
resulting impact on access to cash through the capital markets. 2018 was a record year
for biopharma IPOs, which we believe supports near- to mid-term investment. In light of
market volatility, IPO activity still remained strong in 4Q18, with total deal values >$1.4
billion, up 19% from 4Q17. As a result of elevated capital markets activity in recent years
(2013-2018), aggregate cash levels for the 30 SMiD biopharma companies in our tracker
remain at all-time highs.
45%
52%
52%
46%
43%
56%
70%
72%
78%
89%
77%
71%
91%
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
100%
CDER NME NDAs/BLAs
2
5
6
6
5
2
4
2
3
6
6
6
6
2
11
12
7
12
16
25
19
11
15
31
18
18
16
21
20
15
24
33
25
30
33
15
34
43
26
30
22
26
32
38
26
35
34
36
23
41
41
36
41
35
43
57
43
0
10
20
30
40
50
60
BLA Approvals
NDA Approvals
Filings
剩余44页未读,继续阅读
资源评论


2201_75761617
- 粉丝: 20
- 资源: 7339
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功
