没有合适的资源?快使用搜索试试~ 我知道了~
Springer Handbook of Lasers and Optics 完整版 下
温馨提示

试读
764页
Springer出版社经典光学典籍,该文件是完整版下冊。从理论与应用角度做出了近乎完美的论述,2007年出版。
资源推荐
资源详情
资源评论

563
Coherent
Part C
Part C Coherent and Incoherent Light Sources
10 Incoherent Light Sources
Dietrich Bertram, Aachen, Germany
Matthias Born, Aachen, Germany
Thomas Jüstel, Steinfurt, Germany
11 Lasers and Coherent Light Sources
Orazio Svelto, Milan, Italy
Stefano Longhi, Milano, Italy
Giuseppe Della Valle, Milan, Italy
Stefan Kück, Braunschweig, Germany
Günter Huber, Hamburg, Germany
Markus Pollnau, Enschede, The Netherlands
Hartmut Hillmer, Kassel, Germany
Stefan Hansmann, Darmstadt, Germany
Rainer Engelbrecht, Erlangen, Germany
Hans Brand, Erlangen, Germany
Jeffrey Kaiser, Mountain View, USA
Alan B. Peterson, Mountain View, USA
Ralf Malz, Jena, Germany
Steffen Steinberg, Jena, Germany
Gerd Marowsky, Göttingen, Germany
Uwe Brinkmann, Bovenden, Germany
Dennis Lo
†
, Hong Kong, P. R. China
Annette Borsutzky, Kaiserslautern, Germany
Helen Wächter, Zurich, Switzerland
Markus W. Sigrist, Zurich, Switzerland
Evgeny Saldin, Hamburg, Germany
Evgeny Schneidmiller, Hamburg, Germany
Mikhail Yurkov, Hamburg, Germany
Katsumi Midorikawa, Saitama, Japan
Joachim Hein, Jena, Germany
Roland Sauerbrey, Dresden, Germany
Jürgen Helmcke, Braunschweig, Germany
12 Femtosecond Laser Pulses: Linear Properties,
Manipulation, Generation and Measurement
Matthias Wollenhaupt, Kassel, Germany
Andreas Assion, Vienna, Austria
Thomas Baumert, Kassel, Germany

565
Incoherent Li
10. Incoherent Light Sources
Since the invention and industrialization of
incandescent lamps at the end of the 19th
century electrical lighting has become a com-
modity in our daily life. Today, incoherent
light sources are used for numerous appli-
cation areas. Major improvements have been
achieved over the past decades with respect
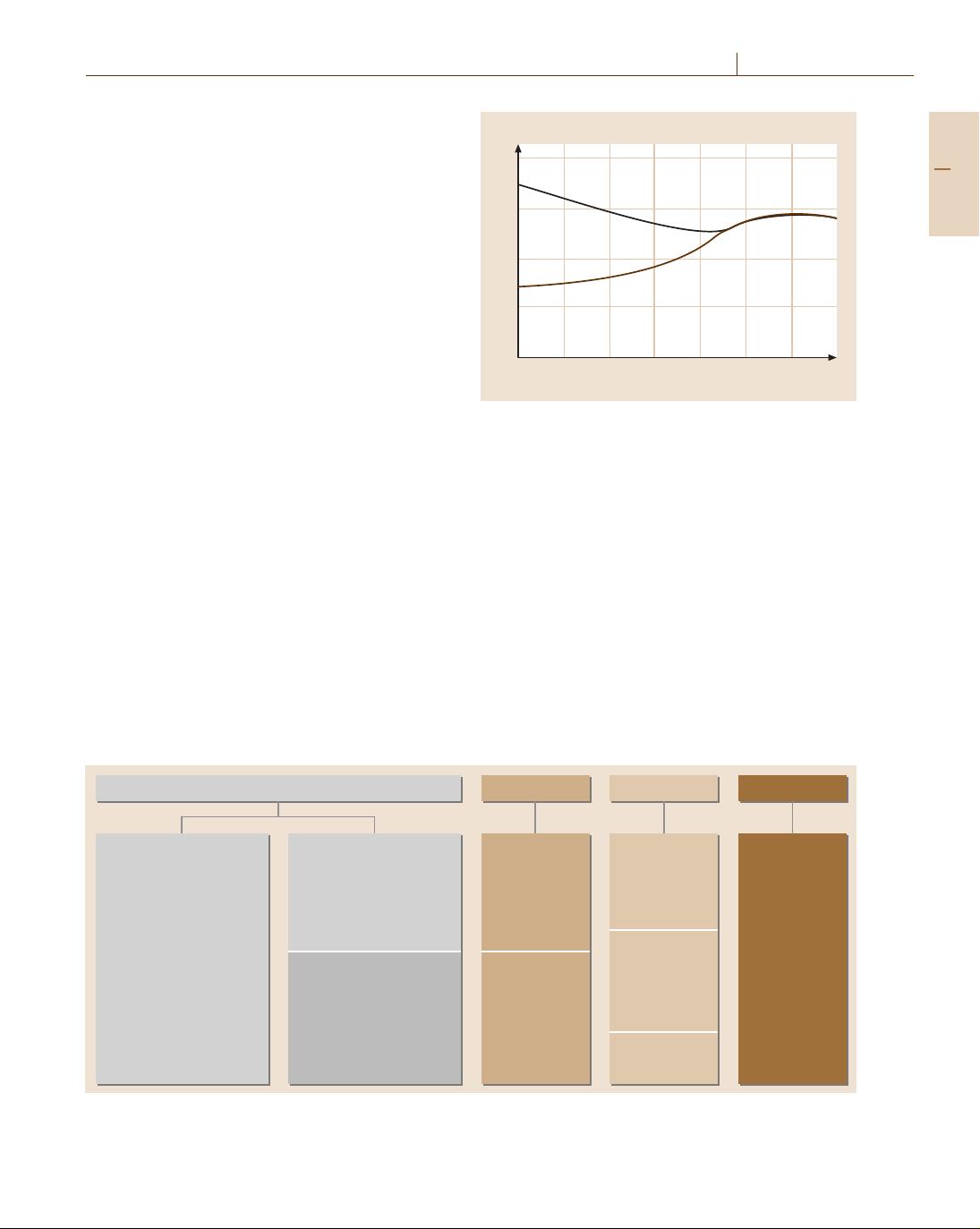
to lamp efficiency Fig. 10.1, lifetime and color
properties.
In the following chapters an overview of var-
ious lamp types and their properties is given.
They are subdivided by light generation mech-
anism: thermal emission of radiation close
to thermal equilibrium (incandescent lamps),
atomic and molecular emission in gas dis-
charge lamps, and emission from solid-state
light sources (LEDs).
10.1 Incandescent Lamps ............................. 565
10.1.1 Normal Incandescent Lamps .......... 565
10.1.2 Tungsten Halogen Lamps .............. 566
10.2 Gas Discharge Lamps ............................ 566
10.2.1 General Aspects ........................... 566
10.2.2 Overview of Discharge Lamps......... 567
10.2.3 Low-Pressure Discharge Lamps ...... 567
10.2.4 High-Pressure Discharge Lamps ..... 570
10.3 Solid-State Light Sources....................... 574
10.3.1 Principle of Electroluminescence .... 574
10.3.2 Direct Versus Indirect
Electroluminescence ..................... 575
10.3.3 Inorganic Light-Emitting Diodes
(LEDs) ......................................... 575
10.3.4 Organic LEDs ................................ 578
10.4 General Light-Source Survey.................. 581
References .................................................. 581
10.1 Incandescent Lamps
The incandescent lamp is the oldest electrical light
source still in widespread use. It can be found in almost
any application, especially where comparatively small
lumen packages are required and where simplicity and
compactness are favored.
10.1.1 Normal Incandescent Lamps
Incandescent lamps produce light by the electrical heat-
ing of a metal wire to such a high temperature that
radiation in the visible part of the spectrum is emit-
ted [10.1]. The metal wire is mounted in a glass bulb
filled with an inert gas (Fig.10.2).
According to Planck’s law the filament must be
heated up to at least 2400 K for a white emission color.
In view of the efficiency for converting electrical en-
ergy into visible light, even higher filament temperatures
would be favorable. Unfortunately, increasing the tem-
perature reduces lamp life due to enhanced evaporation
rates of the metal. Since tungsten has a high melting
point and a low vapor pressure it permits high operating
temperature and consequently higher efficiencies can be
attained than by any other metal. A possible measure to
improve lifetime is the reduction of the tungsten evapo-
rationrate by theaddition of raregases (Kr, Xe).Alterna-
tively, halogens are used in a so-called regenerativecycle
Efficacy (lm/W)
Year
1870
2000
200
175
150
125
100
75
50
25
0
1880
1890
1900
1910
1920
1930
1940 1960
1970
1980
19901950
LP sodium
HP sodium
HP metal halide
LP fluorescent
HP mercury
White LED
Incandescent/Halogen
LP compact fluorescent
Fig. 10.1 Temporal development of the luminous efficacy
of electrical light sources (LP = low pressure, HP = high
pressure)
Part C 10

566 Part C Coherent and Incoherent Light Sources
Fill gas
Filament
Bulb
Support wires
Lead-in wires
Stem
Fuse
Lamp cap
Fig. 10.2 Schematic drawing of an incandescent lamp
effectively to transport tungsten back to the filament. An
increasein luminous efficiencyis achievedby coiling the
tungsten wire. A coiled filament allows higher operation
temperatures for a specified lifetime (typically 1000 h).
Most common operation parameters yield luminous ef-
ficiencies between 8lm/W and 17 lm/Watfilament
temperatures of 2400K and 3100 K, respectively. These
values correspond to an energy efficiency of a few per-
cent. Incandescent lamp wattages range up to 2000W.
Fig. 10.3 Principle of the chemical transport cycle in tung-
sten halogen lamps
10.1.2 Tungsten Halogen Lamps
In a normal incandescent lamp, tungsten evaporates off
the filament and condenses on the bulb wall, result-
ing in so-called blackening. Halogen lamps comprise
a halogen, i. e. iodine, bromine, chlorine, added to the
normal gas filling. These form volatile tungsten com-
pounds at the glass wall, which are transported back
to the hot filament. Here, the tungsten halides are de-
composed enabling a so-called chemical transport cycle
(Fig. 10.3).
By reduction of the net tungsten evaporation rate,
the filament of halogen lamps can be operated at higher
temperature compared to standard incandescent lamps.
Thus, luminous efficiencies can be increased for reduced
lamp size. This allows their application in compact re-
flectors. Halogen incandescent lamps are available up to
2000 W with luminous efficacies up to 25 lm/W. This
value has recently been improved to 35 lm/Wbycoat-
ing the glass bulb with infrared reflective multilayers,
and lifetimes up to 2000h were obtained.
W + 2Br WBr
2
Filament
Bulb wall
W+ 2Br
WBr
2
W
2Br
1500 K
10.2 Gas Discharge Lamps
10.2.1 General Aspects
A gaseous discharge is obtained by driving an
electric current through a gas, typically present
between two electrodes. Alternatively, electrodeless
microwave-exciteddischargesand pulsed dielectric-bar-
rier discharges (e.g. used for plasma displays) are known
as incoherent light sources.
The actual carriers of the electric current in the
gas are electrically charged particles, positive ions and
negative electrons. In a neutral nonconductive gas the
number of charge carrying particles is extremely small.
These particles can be released from the fill gas or the
cathode surface by energetic collisions. Many physical
factors influence the properties of a gas discharge, the
most important ones being the type and pressure of
the gas, the electrode material, the operating tempera-
ture of the electrodes, the shape and surface structure
of the electrodes, the distance between the electrodes,
the geometry of the discharge vessel, and the cur-
Part C 10.2
Incoherent Light Sources 10.2 Gas Discharge Lamps 567
rent density. For the purpose of light generation, two
main types are distinguished: low-pressure and high-
pressure discharge lamps. For lighting applications,
both are operated in the arc discharge mode, which is
characterized by high current densities
> 1A/cm
2
.
To limit the discharge currents, electronic ballasts are
used [10.2].
In a low-pressure discharge lamp (gas pressure typ-
ically less than 100 Pa) the electrons have a mean
free path length larger or of the order of the ves-
sel diameter (e.g. a few cm). Due to low collision
rates with the neutral gas atoms they gain high en-
ergies (> 1 eV) from the applied electrical field and
effectively excite the cold atoms by inelastic collisions.
The electrons and atoms are not in thermal equilibrium
(Fig. 10.4).
In high-pressure discharge lamps the operating pres-
sureis typicallyin therange between 10kPaand 10 MPa.
Here, collisions between electrons and atoms or ions are
much more frequent, resulting in a thermal equilibrium
that is characterized by equal particle temperatures.
In low-pressure discharges atomic line radiation
is emitted preferably from resonance transitions of
the element with the lowest excitation potential (e.g.
Hg: 185 nm and 254 nm, Na: 589 nm). In high-pressure
lamps various contributions to the spectrum are ob-
tained: broadened atomic lines (resonance-, van der
Waals- and Stark-broadening), molecular radiation
bands and quasi-continuous emission due to free–
free (Bremsstrahlung) and free–bound (recombination
of electrons with ions and atoms) transitions. As
a result of these quasi-continuous spectra, the color
Sulphur
High pressure
microwave
S
2
Rare gas
Low pressure
Ne
580 – 720 nm
74 nm
(Phosphors)
p ≈ 0.5 bar
DBD, PDP
Xe/Ne
147 + 172 nm
Phosphors
High pressure
Xe
Sodium
Low pressure
Na/Ar/Ne
Na 589 nm
High pressure
Na/Hg/Xe
High pressure
p > 1bar
Hg/Ar
•p ≈ 20 bar
•p ≈ 200 bar (short arc)
Metal halide lamps
• 3line radiators
NaX/TIX/InX, X = I, Br
• Multi-line/Molecular
NaX/TIX/REX
3
RE = Dy, Ho, Tm, Sc
SnX
2
Mercury
Low pressure
p < 1 mbar
Hg/Ar
Hg/Ne
185 + 254 nm
(Compact)
Fluorescent
lamps
Phosphors
Fig. 10.5 Overview of gas discharge lamps
Temperature (K)
Pressure (Pa)
1
10
5
10
4
10
3
10
2
10
10
–1
10
6
10 10
2
10
3
10
4
10
5
te
tg
Fig. 10.4 Relationship between electron temperature, gas
temperature and gas pressure
rendering properties of high-pressure discharge lamps
are fair to excellent, depending on the type of fill-
ing [10.3].
10.2.2 Overview of Discharge Lamps
In Fig.10.5 the most relevant types of discharge lamps
are displayed. The light sources are distinguished with
respect to their emission spectra and application fields.
In the case of low-pressure mercury and xenon excimer
lamps luminescent materials are applied for conver-
sion of ultraviolet (UV) radiation into visible light.
The temporal improvement in efficiencies is depicted
in Fig. 10.1.
Part C 10.2
568 Part C Coherent and Incoherent Light Sources
10.2.3 Low-Pressure Discharge Lamps
The most widely applied radiators in low-pressure dis-
charge lamps are Hg and Na. These elements are the
best choice with respect to efficiency for the conversion
of electrical input power into radiation.
Low-Pressure Mercury Lamps
The working principle of a low-pressureHg (also known
as a fluorescent lamp) is given in Fig. 10.6. Fluorescent
lamps are generally designed in the form of a linear or
bent tubular bulb with an electrode sealed in at each end
(electrodeless versions are available where the electrical
energy is coupled inductively into the discharge ves-
sel via metal coils). The discharge vessel is filled with
an inert gas (typically Ar) and a few mgHg. Since the
major part of the emission of Hg atoms (97%) at low
pressures (e.g. 5 Pa) is in the ultraviolet, the inner sur-
face of the bulb is coated with a fluorescent powder or
phosphor, which converts the UV radiation into visible
light. The composition of the phosphor determines the
spectral power distribution and the color of the emitted
light.
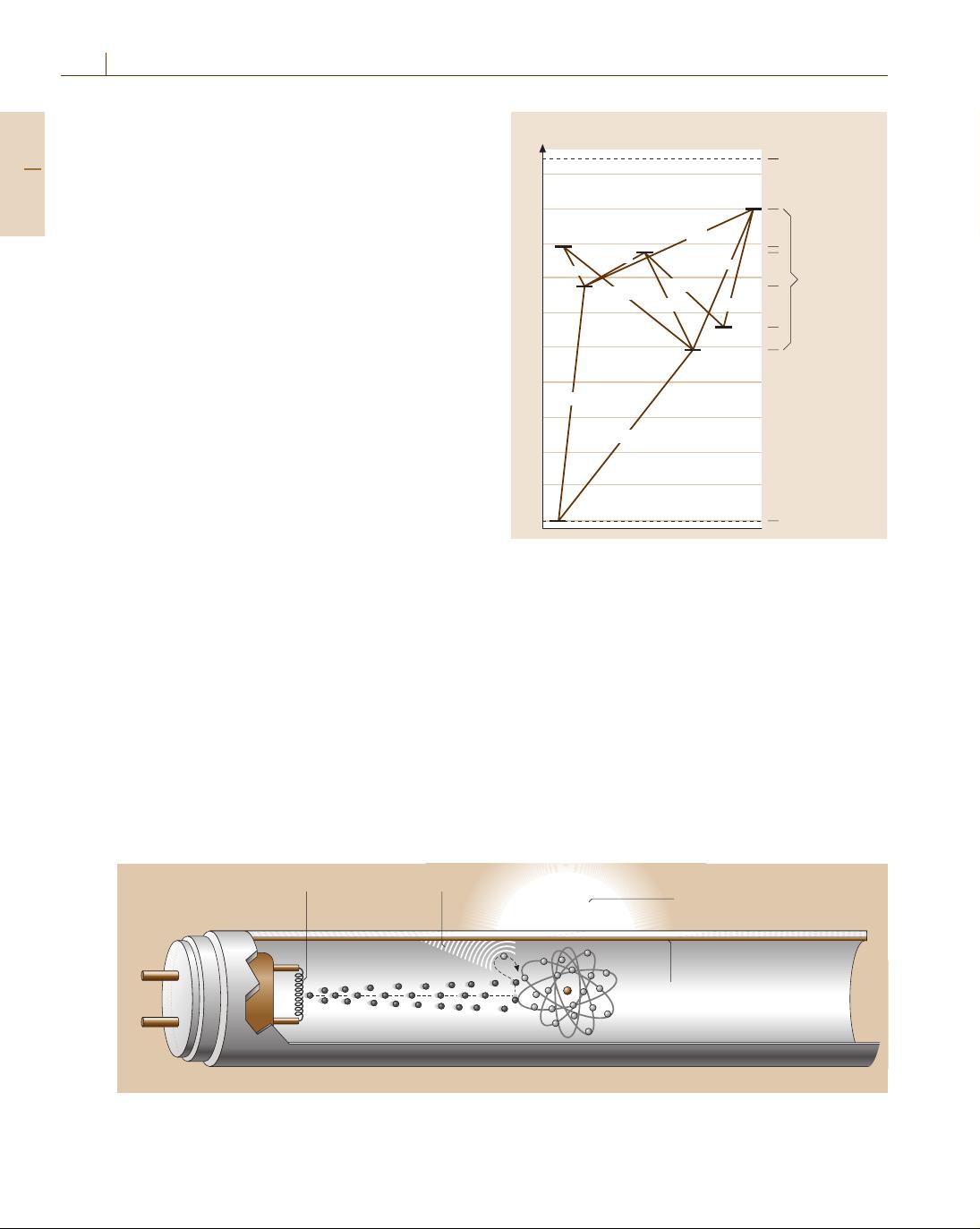
The generation of UV photons is due to transitions of
Hg atoms between the excited state levels
1
P
1
,
3
P
1
and
the ground state level
1
S
0
. About 64% of the electrical
input power is converted into photons at a wavelength
of 185 nm and 254 nm. According to the transitions in-
dicated in Fig. 10.7 only 3% are emitted in the visible
part of the spectrum. As a result of the Stokes shift, the
overall efficiency is only 28%. This value corresponds
to a luminous efficiency of 100lm/W.
Typical lamp parameters for fluorescent lamps are:
electrical input power up to 140 W, luminous efficiency
up to 100 lm/W, color temperature between 2700 K
and 8000 K. Lamps are available in a large variety
Ultraviolet
radiation
Visible radiationElectrode
Electrons Mercury atom
Fluorescent powder
Fig. 10.6 Sketch of a low-pressure mercury discharge (fluorescent) lamp
Energy level above stable level
10
9
8
7
6
5
4
3
2
1
0
Ionization
level
Metastable
level
313 nm
Various
excited
levels
Stable level
366 nm
577 nm
1208 nm
1014
nm
408 nm
436 nm
546 nm
185 nm
254 nm
Fig. 10.7 Simplified energy-level scheme and radiative
transitions of the Hg atom
of geometries, e.g. cylindrical or circular or U-shaped
tubes. The latter are also known as energy-saving or
compact fluorescent lamps. Depending on the con-
struction of fluorescent lamps, the lifetime ranges
from 5000 to 25 000 h.
Fluorescent Coatings
The most important component of a fluorescent lamp is
the fluorescent powder. It is coated onto the inner side
of the glass tube. The powder consists of one or several
luminescent materials (phosphors), which are in general
inorganic compounds doped by transition metals
e.g.
Part C 10.2
剩余763页未读,继续阅读
ratfish0
- 粉丝: 1
- 资源: 4
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功

- 1
- 2
前往页