没有合适的资源?快使用搜索试试~ 我知道了~
Nonlinear interaction decomposition (NID)-A method for separatio...
需积分: 0 0 下载量 99 浏览量
2024-04-05
12:23:11
上传
评论
收藏 2.71MB PDF 举报
温馨提示

试读
14页
Nonlinear interaction decomposition (NID)-A method for separation of cross-frequency coupled sources in human brain.pdf
资源推荐
资源详情
资源评论

Nonlinear interaction decomposition (NID): A method for separation of
cross-frequency coupled sources in human brain
Mina Jamshidi Idaji
a
,
b
,
c
, Klaus-Robert Müller
b
,
d
,
e
, Guido Nolte
f
, Burkhard Maess
j
,
Arno Villringer
a
,
g
, Vadim V. Nikulin
a
,
h
,
i
,
*
a
Department of Neurology, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany
b
Machine Learning Group, Technical University of Berlin, Berlin, Germany
c
International Max Planck Research School NeuroCom, Leipzig, Germany
d
Department of Brain and Cognitive Engineering, Korea University, Anam-dong, Seongbuk-gu, Seoul, Republic of Korea
e
Max Planck Institute for Informatics, Stuhlsatzenhausweg, Saarbrücken, Germany
f
Department of Neurophysiology and Pathophysiology, University Medical Center Hamburg-Eppendorf, Hamburg, Germany
g
Department of Cognitive Neurology, University Hospital Leipzig, Leipzig, Germany
h
Centre for Cognition and Decision Making, Institute for Cognitive Neuroscience, National Research University Higher School of Economics, Moscow, Russia
i
Neurophysics Group, Department of Neurology, Charit
e-Universit
€
atsmedizin Berlin, Berlin, Germany
j
MEG and Cortical Networks Group, Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany
ARTICLE INFO
Keywords:
Nonlinear interaction decomposition
NID
Cross-frequency coupling
MEG
EEG
Nonlinear neuronal interactions
Independent component analysis
ICA
ABSTRACT
Cross-frequency coupling (CFC) between neuronal oscillations reflects an integration of spatially and spectrally
distributed information in the brain. Here, we propose a novel framework for detecting such interactions in
Magneto- and Electroencephalography (MEG/EEG), which we refer to as Nonlinear Interaction Decomposition
(NID). In contrast to all previous methods for separation of cross-frequency (CF) sources in the brain, we propose
that the extraction of nonlinearly interacting oscillations can be based on the statistical properties of their linear
mixtures. The main idea of NID is that nonlinearly coupled brain oscillations can be mixed in such a way that the
resulting linear mixture has a non-Gaussian distribution. We evaluate this argument analytically for amplitude-
modulated narrow-band oscillations which are either phase-phase or amplitude-amplitude CF coupled. We
validated NID extensively with simulated EEG obtained with realistic head modelling. The method extracted
nonlinearly interacting components reliably even at SNRs as small as 15 dB. Additionally, we applied NID to the
resting-state EEG of 81 subjects to characterize CF phase-phase coupling between alpha and beta oscillations. The
extracted sources were located in temporal, parietal and frontal areas, demonstrating the existence of diverse local
and distant nonlinear interactions in resting-state EEG data. All codes are available publicly via GitHub.
1. Introduction
Oscillatory neuronal activity has been associated with almost all brain
operations including sensory, motor and cognitive processes (Buzs
aki
and Draguhn, 2004). In humans, these oscillations can be measured with
magneto- and electroencephalography (MEG/EEG), where the frequency
content is classically divided into specific frequency bands, namely δ
(0.5–4 Hz), θ (4–8 Hz),
α
(8–12 Hz), β (12–25 Hz), γ (25–70 Hz).
1
Each
frequency band has been associated with specific functional roles. For
example, alpha oscillations are known to be relevant for attention/sen-
sory processing (Groppe et al., 2013; Klimesch, 2012), while beta-band
activity is primarily associated with sensorimotor processing (Bayr-
aktaroglu et al., 2011; Kilavik et al., 2013; Klimesch, 2012; Salmelin and
Hari, 1994). While specific neuronal operations can be carried out by
oscillations in distinct frequency bands, there should be neuronal
mechanisms integrating such spatially and spectrally distributed pro-
cessing (Palva et al., 2005). In this way, neuronal communications can be
considerably enriched via coupling of neuronal oscillations within one
frequency band (Engel and Fries, 2010; Fries, 2015) as well as between
different frequency bands. Various types of cross-frequency (CF) in-
teractions among neural oscillations, namely phase-phase, amplitu-
de-amplitude, phase-amplitude coupling have been observed in human
* Corresponding author. Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig, Germany.
1
The range of frequencies in each frequency band slightly differs in different references.
Contents lists available at ScienceDirect
NeuroImage
journal homepage: www.elsevier.com/locate/neuroimage
https://doi.org/10.1016/j.neuroimage.2020.116599
Received 25 November 2019; Received in revised form 16 January 2020; Accepted 31 January 2020
Available online 5 February 2020
1053-8119/© 2020 The Authors. Published by Elsevier Inc. This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-
nc-nd/4.0/).
NeuroImage 211 (2020) 116599

electrophysiological recordings (e.g. MEG/EEG) (Canolty and Knight,
2010; Jensen and Colgin, 2007; Nikulin and Brismar, 2006; Palva et al.,
2005) and have been linked to diverse perceptual and cognitive processes
(Canolty and Knight, 2010; Fell and Axmacher, 2011; Hyafil et al., 2015;
Palva et al., 2005; Sauseng et al., 2008; Siebenhühner et al., 2016). In this
study, we focus on the extraction of these interactions from
multi-channel MEG/EEG. While the novel approach introduced here is
applicable to different types of CFC, a special emphasis is dedicated to
phase-phase coupling for the following reasons.
The phase of neuronal oscillations is known to represent the timing of
the firing of a neuronal population generating the oscillation (Fries,
2009, 2015; Palva et al., 2005; Siegel et al., 2012), while its amplitude
reflects the strength of local spatial synchronization (Siegel et al., 2012).
The interaction of the activities of distinct neuronal populations is
manifested in the locking of phase/amplitude of the observed oscilla-
tions. Phase-phase coupling is a type of CFC that operates with milli-
second precision for both oscillations (Fell and Axmacher, 2011; Marzetti
et al., 2019; Palva et al., 2005; Siegel et al., 2012) and investigating it
with MEG/EEG recordings can provide a unique possibility to study
synchronization of the spiking of distinct neuronal populations
non-invasively (Palva and Palva, 2018).
A number of previous studies have investigated CF phase synchro-
nization in sensor-space (Darvas et al., 2009; Nikulin and Brismar, 2006;
Palva et al., 2005; Tass et al., 1998). However, volume conduction does
not allow the disentanglement of individual components. In order to
resolve this issue, some previous studies have investigated the phase
synchrony in the source-space using inverse modelling (Siebenhühner
et al., 2016; Tass et al., 2003). Yet, source-space analysis is computa-
tionally exhausting and source reconstruction methods are ill-posed,
which may lead to inconsistent outcomes (Mahjoory et al., 2017). On
the other hand, due to a linear mapping of the neuronal source signals to
the sensors, multivariate methods can increase the signal-to-noise ratio
(SNR) and accuracy of localizing the neuronal activity (Parra et al.,
2005). At the same time, these methods alleviate the problem of multiple
testing in sensor- or source-space analysis. While most of the multivariate
source-separation methods focus on the extraction of independent sour-
ces (e.g. independent component analysis - ICA), there are only a few
studies utilizing multivariate methods to extract dependent sources from
the electrophysiological recordings of the human brain (Chella et al.,
2016;
Cohen, 2017; D
€
ahne et al., 2014; Nikulin et al., 2012; Volk et al.,
2018). These methods optimize a contrast function of the desired type of
coupling. However, we show that the coupling can be reflected in the
statistical properties of the signal constructed through the linear mixing
of nonlinearly coupled processes. We refer to our method as Nonlinear
Interaction Decomposition (NID).
The rest of the manuscript is organized as follows. In section 2 we
provide some preliminary background about the amplitude-modulated
narrowband oscillations and their linear mixture. Section 3 is dedi-
cated to explaining the proposed method (NID) and its algorithmic steps.
In section 4 the experimental data and the analysis/testing approaches
are described. The results of applying NID to simulated as well as resting-
state EEG data are presented in section 5. Finally, a discussion and a
conclusion are provided in the last section.
2. Preliminary background
In this section we introduce the main assumptions and the core idea of
NID.
2.1. Nomenclature
We start with defining the key phrases used throughout the manu-
script. An amplitude-modulated (AM) signal is a signal with a non-
constant envelope. An amplitude-modulated narrow-band (AM narrow-
band) signal is an AM signal whose energy is concentrated in a specific
narrow bandwidth. For instance, alpha-waves in M/EEG are AM narrow-
band signals, whose energy is in the bandwidth of 8 12 Hz.
2.2. Linear mixture of cross-frequency coupled brain oscillations
In order to understand the idea of NID, it is helpful to consider non-
sinusoidal brain oscillations. Note that NID does not require oscilla-
tions to be non-sinusoidal, they are rather used here for the demonstra-
tion of the method. The frequency content of such signals is concentrated
at two (or more) narrow bandwidths, whose central frequencies are
multiples (known as harmonic frequencies (Oppenheim et al., 1983)) of
the fundamental frequency. This means that such a non-sinusoidal signal
can be decomposed to narrow-band components which are phase-phase
coupled to each other. Fig. 1 depicts an example of this observation in
real data. Interpreting a non-sinusoidal signal as a linear mixture of
narrow-band phase-coupled signals, led to the idea that the linear
mixture of nonlinearly coupled narrow-band oscillations has a
non-Gaussian distribution, regardless of the location of the oscillation.
Fig. 2 illustrates an example from real data, where two signals in alpha
and beta frequency band are phase-coupled to each other and their linear
mixture is more non-Gaussian than each of them. Supplementary code
(2) provides some simulations for further illustration of NID’s core idea.
We assume that the distribution of AM narrow-band brain oscillations
do not deviate strongly from Gaussian distribution. It is also discussed in
(Hyv
€
arinen et al., 2010) that the amplitude modulation of brain oscil-
lations is the key property that results in the observation that the dis-
tribution of AM narrow-band oscillations does not deviate strongly from
Gaussian distribution. Note that a sufficient amount of data points is
needed so that the histogram of data can become a fair estimation of its
distribution. For example, if the signal is filtered with a very narrow
band-pass filter, more data points are needed to capture the fluctuations
of the amplitude modulation compared to filtering with a broader
band-pass
filter.
Our proposed method (nonlinear interaction decomposition-NID) is
based on the idea that if two narrow-band oscillations are independent or
only linearly coupled, the distribution of their linear mixture is closer to
Gaussian distribution in comparison to the distributions of linear mix-
tures of nonlinearly coupled oscillations. We have analytically proved
that CFC phase-phase and amplitude-amplitude coupled AM narrow-
band oscillations can be linearly mixed to a non-Gaussian distributed
signal (supplementary text, section 1) Fig. 3 illustrates the principle of
NID. Note that we assess the non-Gaussianity of a random variable by
means of kurtosis, skewness, or fifth order moment, all of which are zero
for Gaussian random variables.
3. Method
3.1. Notation
We use boldface lower-case letters (e.g. x) to denote vectors, while
boldface capital letters (e.g. X) are used for matrices. Regular letters, (e.g.
x), indicate scalars. Vectors are used to denote the time series of a signal
or spatial filters/activation patterns. Matrices are used to denote the
concatenation of vectors. The operators ½:; : and ½:; : stand for horizontal
and vertical concatenation of two matrices respectively.
3.2. Measuring cross-frequency coupling
Depending on the type of the coupling, there are different measures to
quantify CFC. In this paper, we worked with phase-phase and amplitude-
amplitude coupled oscillations. As described below, the phase locking
value (PLV) was used for measuring phase-phase coupling, while
amplitude-amplitude coupling was quantified with the envelope corre-
lation. Both of these measures are calculated from the instantaneous
phase and amplitude of oscillations, which are computed as the phase
and magnitude of the complex analytic signal based on the Hilbert
transform.
M.J. Idaji et al. NeuroImage 211 (2020) 116599
2
Phase-phase coupling. Oscillations with frequencies f
n
and f
m
¼
m
n
f
n
; n;
m 2 ℕ are called n:m phase-coupled if jmΦ
n
ðtÞnΦ
m
ðtÞj < const , where
Φ
n
ðtÞ and Φ
m
ðtÞ define the instantaneous phases of the two oscillations at
f
n
and f
m
respectively. To quantify n : m phase-phase coupling, phase-
locking value (PLV) is widely used (Palva et al., 2005; Sauseng et al.,
2008; Scheffer-Teixeira and Tort, 2016; Siebenhühner et al., 2016) and it
is defined as
< e
jΨ
n;m
ðtÞ
>
, where Ψ
n;m
ðtÞ¼ðmΦ
n
ðtÞnΦ
m
ðtÞÞ, <:>
stands for computation of the mean over time samples, j is the imaginary
number, and j:j is the absolute value operator.
Amplitude-amplitude coupling. In the case of amplitude-amplitude
coupling, the instantaneous amplitudes of oscillations are correlated.
Therefore, the correlation coefficient of the oscillations’ envelopes in-
dicates the strength of the amplitude-amplitude coupling.
3.3. Detection of cross-frequency coupling: problem formulation
We assume that there are N non-linearly coupled pairs of source
signals
fð
s
ðnÞ
i
; s
ðmÞ
i
Þg
N
i¼1
at frequencies f
n
and f
m
, where f
n
¼ nf
b
and f
m
¼
mf
b
. f
b
is a base-frequency relating f
n
and f
m
to each other. In the rest of
the paper, all the criteria and equations mentioned for frequency f
n
holds
for frequency f
m
as well. s
ðnÞ
i
2 R
1T
is a narrow-band source signal at f
n
,
where T is the number of time samples. The electrical (or magnetic) ac-
tivity measured at the sensors can be modeled as a linear mixture of the
sources as in the following (Baillet et al., 2001; Haufe et al., 2014):
x ¼ P
ðnÞ
S
ðnÞ
þ P
ðmÞ
S
ðmÞ
þ ξ (1)
where X 2 R
CT
is the matrix of multi-channel measured signal with C as
the number of channels. P
ðnÞ
¼½
p
ðnÞ
1
; ⋯; p
ðnÞ
N
. We call p
ðnÞ
i
2 R
C1
the
mixing pattern of source s
ðnÞ
i
. Additionally, S
ðnÞ
¼
½
s
ðnÞ
1
; ⋯; s
ðnÞ
N
2 R
NT
is
the matrix of source signals at f
n
, which are CF coupled to sources in
matrix S
ðmÞ
¼½
s
ðmÞ
1
; ⋯; s
ðmÞ
N
. In equation (1), ξ denotes the noise signal,
which cannot be explained by the linear model. Note that the superscript
of the variables is an indication of their frequency, e.g the superscript ðnÞ
in s
ðnÞ
i
is related to the subscript n of f
n
. As mentioned in section 3.2, the
coupling is called n : m coupling if ðs
ðnÞ
i
; s
ðmÞ
i
Þ are phase-phase coupled.
However, we use this notation for amplitude-amplitude coupling as well
so that we can denote the frequency ratios easier.
We provide an example here. Assume that we have two coupled
source signals in
α
and β frequency band, i.e. N ¼ 2, n ¼ 1; m ¼ 2, and
f
b
¼ 10 Hz; f
1
¼ 10 Hz; f
2
¼ 20 Hz. Then S
ð1Þ
¼
s
ð1Þ
1
; s
ð1Þ
2
and S
ð2Þ
¼
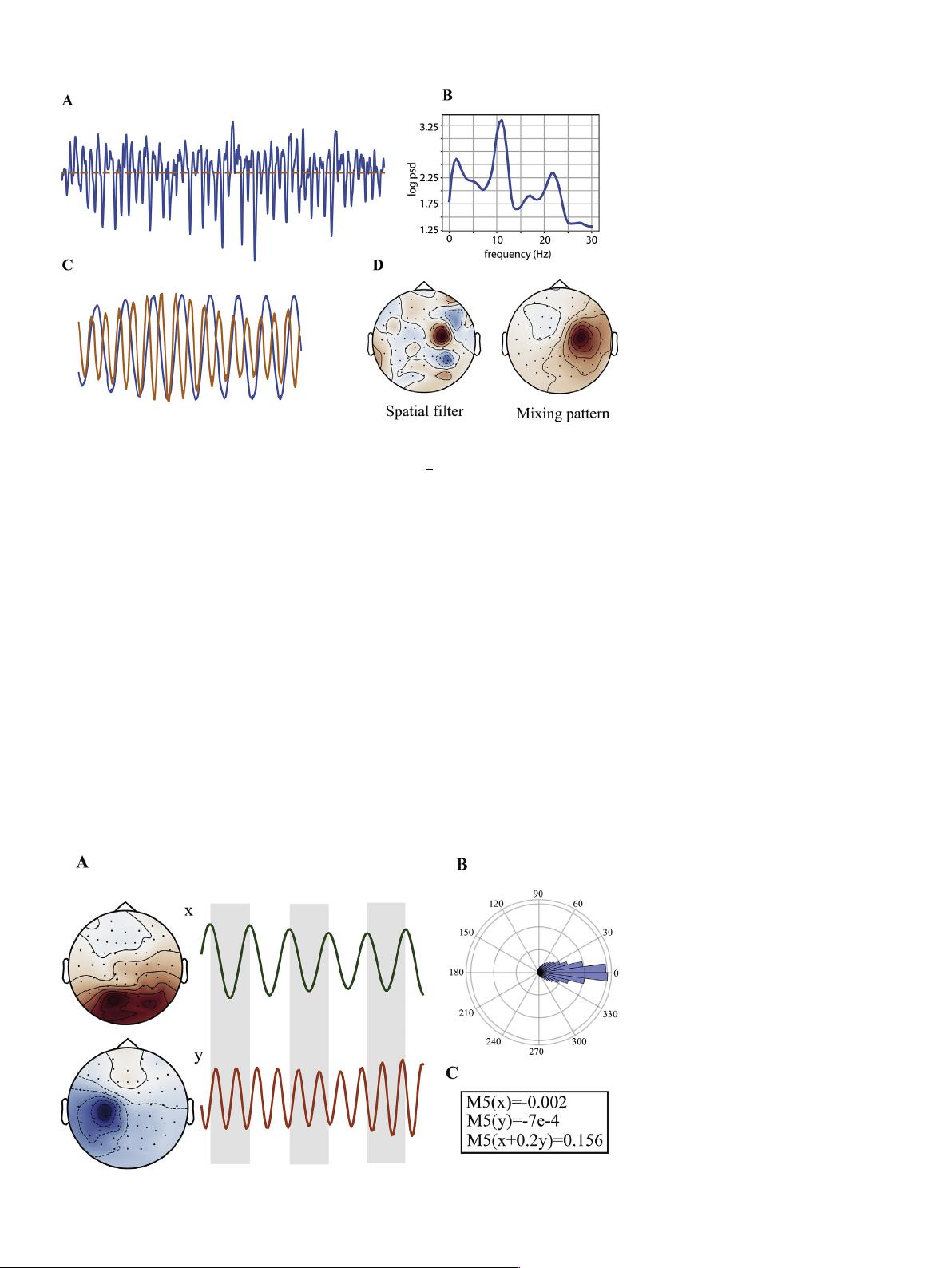
Fig. 1. A non-sinusoidal oscillation obtained from
spatial filtering of EEG of a subject of LEMON
dataset (Babayan et al., 2019) with the spatial filter
in panel D. Panel (A) shows a segment of the time
series of the oscillation with a power spectral
density (PSD) shown in panel (B). The PSD of the
oscillation has clear peaks in alpha and beta bands.
Panel (C) shows a segment of the narrow-band
components (alpha and beta) of the oscillation in
(A). The two components are phase-coupled. Panel
(D) depicts the spatial filter and mixing pattern
(Haufe et al., 2014) of the oscillation, computed
with NID.
Fig. 2. Two phase coupled sources in alpha (x) and
beta (y) band extracted with NID using real EEG
data of a subject from LEMON dataset (Babayan
et al., 2019). Panel (A) shows a segment of alpha
and beta oscillations and their spatial patterns.
Panel (B) depicts the histogram of 2Φ
x
Φ
y
, where
Φ stands for the phase of a signal. The fact that the
phase difference is located in a small sector of the
phase diagram indicates a strong coupling between
alpha (x) and beta (y) oscillations. Panel (C) shows
the value of the fifth moment (denoted as M5) of
the narrow-band signals in panel (A) and their
linear mixture, indicating more non-Gaussianity for
the mixture than for the constituent oscillations.
Note that the fifth moment is used as a measure of
non-Gaussianity in NID’s algorithm.
M.J. Idaji et al. NeuroImage 211 (2020) 116599
3
剩余13页未读,继续阅读
资源评论

2401_83389103
- 粉丝: 0
- 资源: 1
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功
