Sound absorption based on ocean measurements. Part II: Boric acid
contribution and equation for total absorption
R.E. Francois and G. R. Garrison
Applied Physics Laboratory, University of Washington, 1013 N. E. 40th Street, Seattle, Washington 9810.5
{Received 15 June 1982; accepted for publication 18 August 1982}
The absorption of sound in seawater is considered as the sum of three contributions: those from
pure water, magnesium sulfate, and boric acid. Contributions from other reactions are small and
are not included. The pure water and magnesium sulfate contributions obtained from analyses of
extensive oceanic measurements, including many in the Arctic, were discussed in Part I. In Part
II, an analysis is made of all reported measurements in the low-frequency region (0.2-10 kHz) to
evaluate the contribution of boric acid. This is done by subtracting the pure water and magnesium
sulfate contributions determined in Part I from the total absorption to give a more accurate
estimate of the boric acid contribution than previously obtained. The three contributions are then
combined to form an equation with both a theoretical basis and a satisfactory empirical fit that
will be useful to researchers and engineers in the field of underwater sound. The equation applies
to all oceanic conditions and frequencies from 200 Hz to 1 MHz.
PACS numbers: 43.30.Bp, 92.10.Vz, 43.35.Bf, 43.35.Fj
INTRODUCTION
The general equation for the absorption of sound in
seawater gives the absorption as the sum of contributions
from boric acid, magnesium sulfate, and pure water:
A•P•f•f 2 A2P:f:f •
O• = -•- -•- A 3P3 f2, (1)
f• _+_f2 f• _+_f2
wheref is the frequency of the sound, fl and f2 are the relaxa-
tion frequencies of boric acid and magnesium sulfate, and P1,
P2, and P• are nondimensional pressure correction factors.
The measurements and analyses reported earlier in Part
I1 resulted in more accurate equations for the pure water and
magnesium sulfate contributions. Part II treats the boric
acid contribution, which is important at frequencies below
about 10 kHz.
Numerous low-frequency measurements of absorption
have been made in various oceans using sound produeed by
explosives and transmitted along a sound channel. In ana-
lyzing these measurements, other investigators have gener-
ally treated the boric acid contribution as a relaxation pro-
cess, but have usually accepted 1 kHz as the relaxation
frequency regardless of the water properties. Our approach
was to subtract the two components that were delineated in
Part I from the total absorption to obtain a better measure of
the boric acid portion. With three adjustable parameters
available--the boric acid relaxation frequency, the absorp-
tion at that frequency, and a frequency-independent bias in a
{such as a spreading loss error}rowe made a three-variable
least-squares fit to each set of data and obtained a relaxation
frequency that varies with temperature and a proportional-
ity factor that varies with pH.
In this paper, we first summarize the measurements re-
ported by others for which the acoustic and environmental
data were sufficiently documented to use in our analysis. We
then analyze five selected sets whose frequencies bracketed
the relaxation frequency to determine equations for the re-
laxation frequency and the absorption, and compare the bo-
ric acid equation thus derived with the remaining absorption
measurements.
The paper concludes by combining the three contribu-
tions into a single equation and comparing it with the equa-
tions of other investigators. A one-page table of absorption
versus frequency and temperature is included for the
reader's convenience.
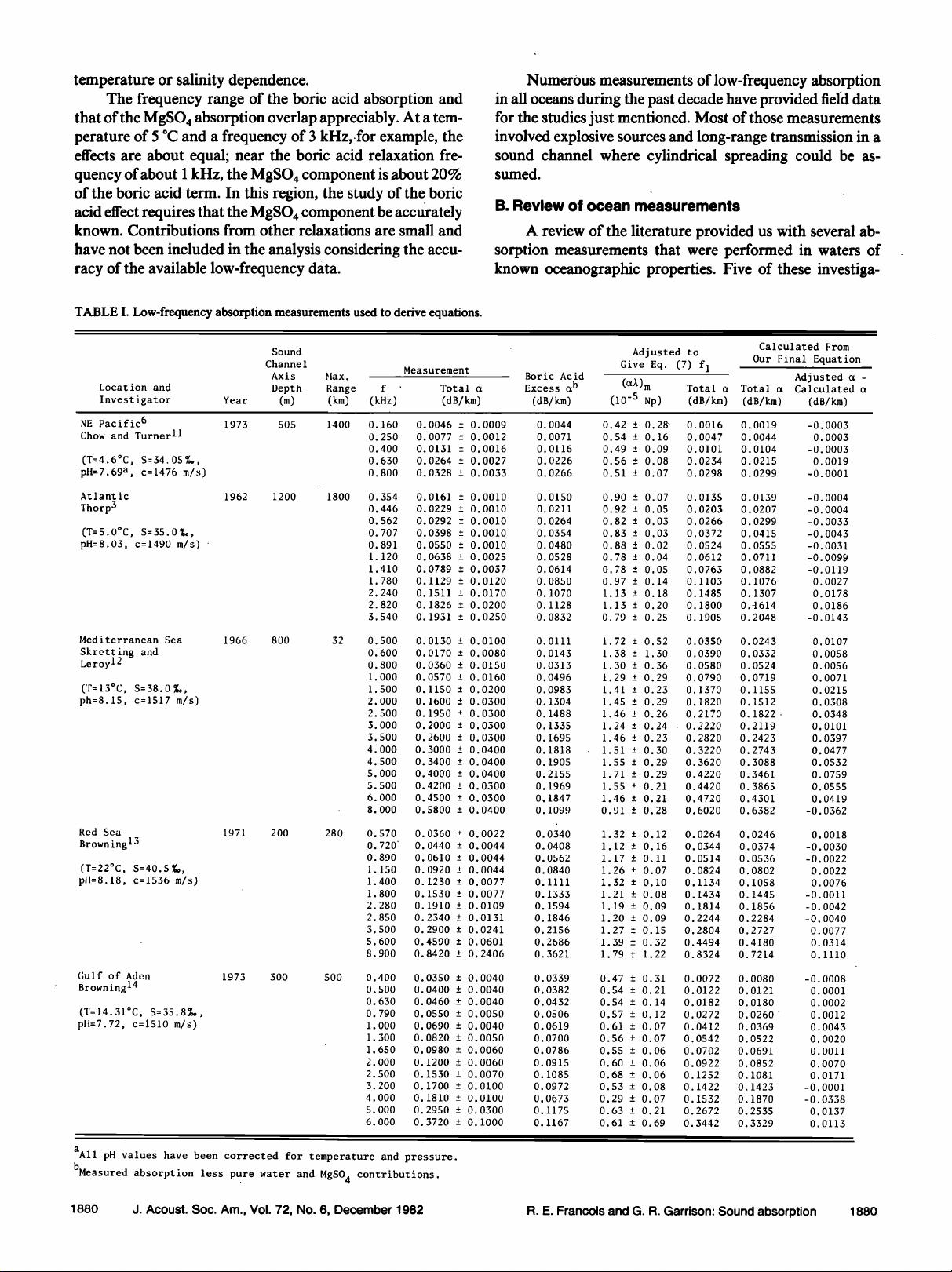
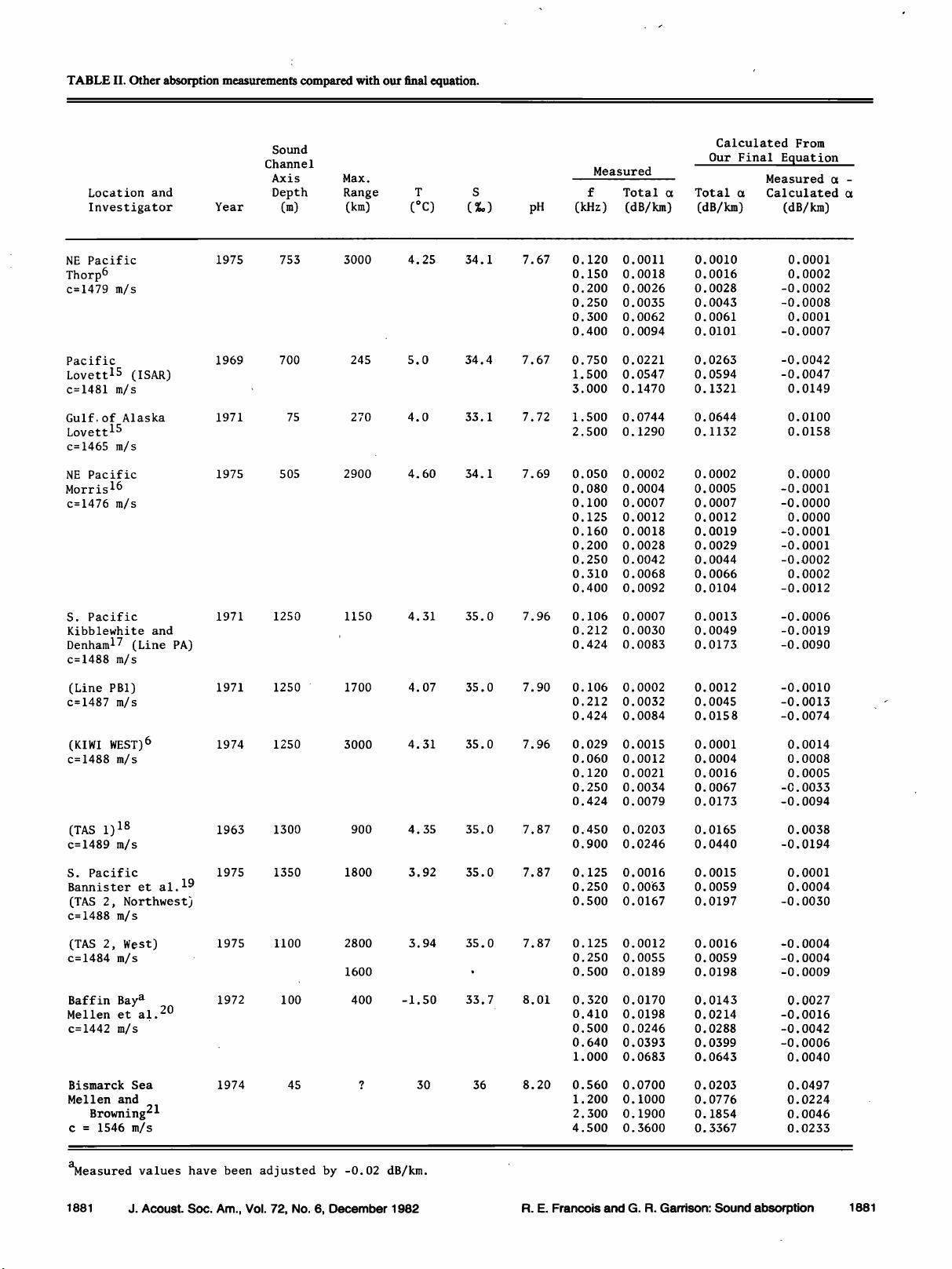
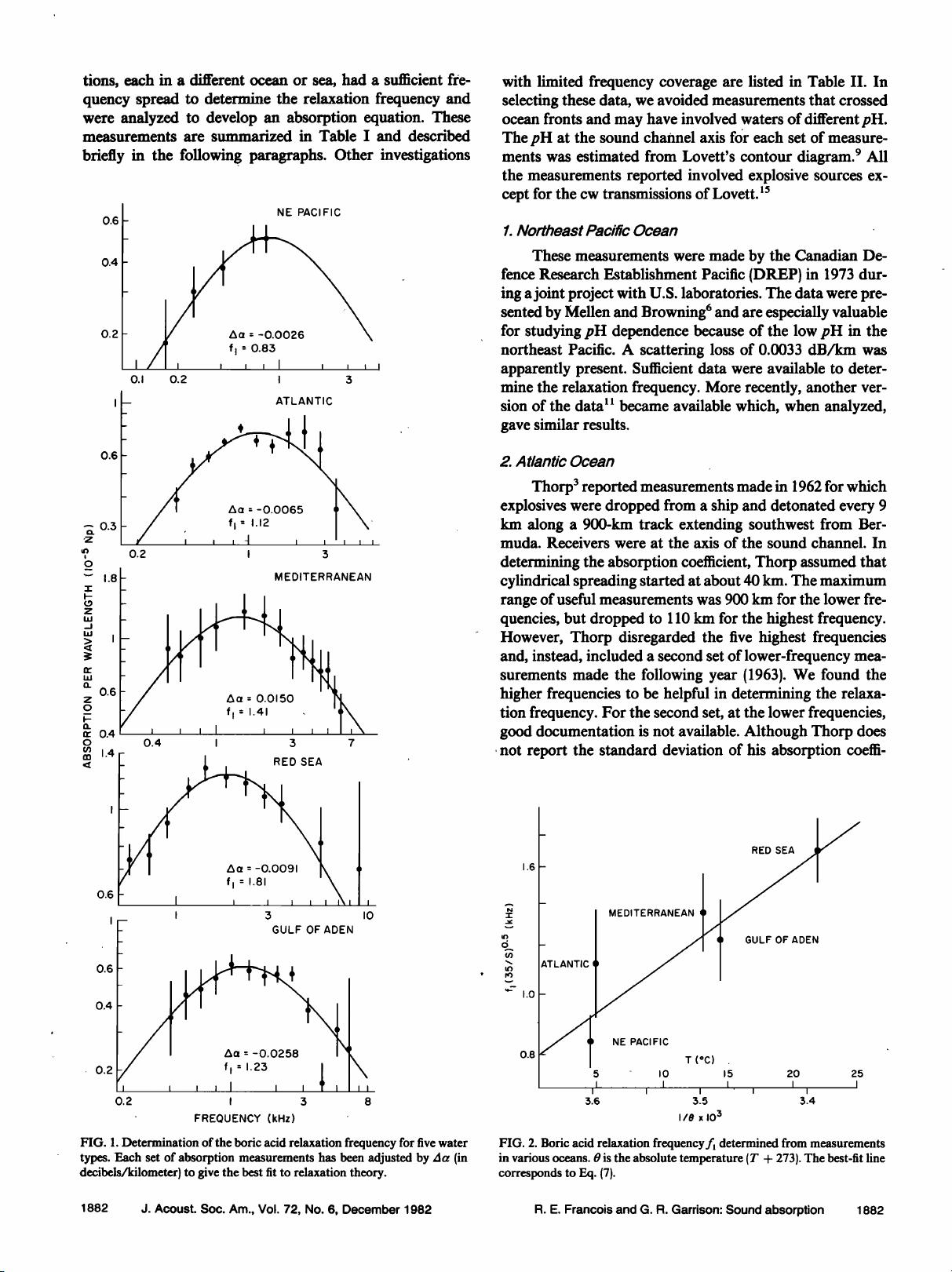
I. LOW-FREQUENCY MEASUREMENTS
A. Absorption anomaly
Measurements of low-frequency absorption in the
1950's showed higher values than expected from pure water
and magnesium sulfate. Sehulkin 2 proposed a nonchemical
relaxation mechanism. A summary by Thorp 3 showed a pre-
ponderance of data that indicated an anomaly below 10 kHz.
In 1972 this anomaly was shown by Yeager et al. 4 to be
caused mainly by a chemical relaxation of boric acid. Sim-
mons, 5 using laboratory measurements, verified this and
noted a variation between the Atlantic and Pacific Oceans.
Mellen and Browning 6 reported that boron-induced absorp-
tion in the Pacific Ocean was about half that in the Ariantie.
Further comparisons by Mellen and Browning 7 of the ab-
sorption and water properties in several widely spread loca-
tions showed that the absorption varied with pH.
At about the same time, Schulkin and Marsh 8 also ex-
amined the ocean variations reported by Simmons. They es-
timated the pH at the sound channel axis for each area, and
also found a strong correlation between absorption and pH.
Following Glotov's theory, they related the relaxation fre-
quency of the boron absorption to the viscosity of the
seawater as well as to the temperature and salinity, and pub-
lished an equation for the boric acid contribution to absorp-
tion. Lovett, 9 using a Russian oceanographic atlas •ø to ob-
tain pH values for the Atlantic, Indian, and Pacific Oceans,
published a world contour map for predicting low-frequency
absorption at the sound channel axis but gave no specific
1879 J. Acoust. Soc. Am. 72(6), December 1982 1879
Redistribution subject to ASA license or copyright; see http://acousticalsociety.org/content/terms. Download to IP: 222.240.177.34 On: Thu, 23 Jul 2015 01:58:29