没有合适的资源?快使用搜索试试~ 我知道了~
镉是一种已知的针对各种器官的环境污染物。 镉毒理学经常牵涉到活性氧的形成,使自由基清除机制不堪重负并引起氧化应激。 急性镉中毒已显示可降低抗氧化酶的活性并诱导氧化应激。 但是,慢性中毒会使氧化应激的结果模糊不清,而细胞会进行调整以克服有毒物质的负荷。 炎症也与氧化应激的发生有关。 急性或慢性炎症的刺激由不同的级联介导。 但是,关键事件包括转录因子,NF-κB的激活和促炎性细胞因子的释放。 在镉暴露下,氧化应激和炎症同时涉及发病机理和多器官组织损伤的诱导。 本文回顾了急性和慢性镉中毒对诱导氧化应激,炎症从而造成组织损伤的影响。
资源推荐
资源详情
资源评论

Occupational Diseases and Environmental Medicine, 2019, 7, 144-163
https://www.scirp.org/journal/odem
ISSN Online: 2333-357X
ISSN Print: 2333-3561
DOI:
10.4236/odem.2019.74012 Oct. 17, 2019 144
Occupational Diseases and Environmental Medicine
Cadmium Toxicity: Oxidative Stress,
Inflammation and Tissue Injury
Sandra Concepcion Das
1,2*
, Hamda A. Al-Naemi
1,2*
1
Laboratory Animal Research Center, Qatar University, Doha, Qatar
2
Department of Biological & Environmental Sciences, Qatar University, Doha, Qatar
Abstract
Cadmium is a known environmental pollutant targeting various organs. Of-
ten implicated in cadmium toxicology is the formation of reactive oxygen
species, overwhelming the free radical scavenging mechanisms and inducing
oxidative stress. Acute cadmium intoxication has been shown to reduce anti-
oxidant enzyme activity and induce oxidative stress. However, chronic intox-
ication has obscure outcomes in oxidative stress while the cell makes adjust-
ments to overcome the toxicant load. Also linked with the occurrence of
oxidative stress is inflammation. Stimulation of acute or chronic inflamma-
tion is mediated by different cascades. However, key events include activation
of transcription factor, NF-
κ
B and release of pro-
inflammatory cytokines.
Both oxidative stress and inflammation are implicated simultaneously in pa-
thogenesis and induction of multi-organ tissue damage under cadmium ex-
posure. This article reviews the impact of acute and chronic cadmium intoxi-
cation on inducing oxidative stress, inflammation and thereby inflicting tis-
sue damage.
Keywords
Cadmium, Inflammation, Tissue Injury
1. Cadmium as an Environmental Pollutant
Cadmium is naturally occurring element in the Earth’s crust with concentrations
of 0.1 - 0.5 ppm in association with ores of zinc, lead and copper. It is a heavy
metal that has various industrial applications with about 24,000 metric tons be-
ing produced yearly worldwide [1]. This production mainly caters to the manu-
facturing of nickel-cadmium batteries, pigments, chemical stabilizers, metallic
coatings and alloys. In the environment, Cd is derived naturally and anthropo-
How to cite this paper:
Das,
S.C. and
Al
-Naemi, H.A. (2019)
Cadmium Toxicity:
Oxidative Stress, Inflammation and Tissue
Injury
.
Occupational Diseases and Env
i-
ronmental
Medicine
,
7
, 144-163.
https://doi.org/10.4236/odem.2019.74012
Received:
August 25, 2019
Accepted:
October 14, 2019
Published:
October 17, 2019
Copyright © 201
9 by author(s) and
Scientific
Research Publishing Inc.
This work is licensed under the Creative
Commons Attribution International
License (CC BY
4.0).
http://creativecommons.org/licenses/by/4.0/
Open Access

S. C. Das, H. A. Al-Naemi
DOI:
10.4236/odem.2019.74012 145
Occupational Diseases and Environmental Medicine
genically, frequently circulating between three environmental compartments of
the air, water and soil. A contributing factor to its circulation in the environ-
mental compartments is its high persistence due to non-biodegradability. Addi-
tionally, the metal is reported to have a high rate of transfer from soil-to-plant,
making it possible for Cd to enter the food chain [2].
Cadmium is considered to be a xenobiotic metal as it is known to be toxic to
plants, animals and humans even at low concentrations and has no essential bi-
ological functions [3]. Owing to its ubiquity, toxicity and long biological half-life
(ranging between 10 - 35 years) [4], its rising levels in the environment are cause
for public health concern. For humans, Cd can enter the body by inhalation of
tobacco smoke, ingestion of contaminated food and water, dermal absorption
and occupational exposure. For non-occupationally exposed and non-smokers,
the main route of exposure is via ingestion of Cd-contaminated food and water.
The route of exposure impacts Cd absorption wherein inhalation contributes of
up to 50%, ingestion contributes to an estimated 10% and is nearly negligible by
dermal contact [5]. The ingested amount can increase up to 20% based on the
nutritional status of the individual [4]. Despite its industrial importance, recent-
ly, it is recognized as one among the 126 priority pollutants [6], a Class I carci-
nogen [7] and is considered an environmental pollutant and health toxicant [5]
[8].
Tissue Cd concentrations
vary based on the route, duration and dose of expo-
sure. The primary sinks of Cd body burden are stored in the kidney and liver
ranging from 50% - 85% out of which 30% - 60% is stored in the kidney alone.
Recently, it has been estimated that dietary Cd exposure contributed to 0.2% of
the global burden of chronic kidney disease [9]. Apart from the primary stores,
Cd-induced damage
has also been reported in brain [10], cardiovascular system
[11] [12], nervous system [13], immune system [14], gut [15] and reproductive
system [16] [17]. In addition to its ability to bioaccumulate, a relatively small
fraction (0.007%) of Cd is excreted from the body as urine and feces [4]. A well
cited epidemiological example of Cd-induced intoxication is the case of the
“Itai-itai” disease in Japan first identified in the 1960s and Cd-associated mortal-
ity is reported till date [18]. Various reviews discuss the oxidative mechanisms
and the inflammatory properties of incited by Cd exposure separatelyhowever
there is accumulating evidence of the interplay between oxidative stress and in-
flammation [19] [20] [21]. This review aims to provide an overview on the link
between Cd-induced oxidative stress, Cd-induced inflammation while discussing
its association with inflicted tissue injury as a result of Cd toxicity.
2. Cadmium-Induced Oxidative Stress
Pathologies of Cd intoxication has been associated with promoted oxidative
stress [22]. Oxidative stress is characterized by an imbalance in the production
of oxidants and antioxidants in the organ or the organism, favoring the former
and causing cellular disruption [23]. It has been discussed that this imbalance
may be a result of either overproduction of reactive oxygen species (ROS) and
S. C. Das, H. A. Al-Naemi
DOI:
10.4236/odem.2019.74012 146 Occupational Diseases and
Environmental Medicine
reactive nitrogen species (RNS) or weakening of the elimination of ROS by the
oxidant defense mechanisms [24]. However, physiological amounts of ROS are
required for normal cellular functions such as signal transduction, cell prolifera-
tion, gene expression and immune defense. Reactive oxygen species consist of
various amounts of hydrogen peroxide (H
2
O
2
), hydroxyl radical (HO•), supe-
roxide anions (
2
O
−
), peroxyl (RO•) and alkoxyl radicals (ROO•) whereas reac-
tive nitrogen species (RNS) include nitric oxide radical (NO•), nitrogen dioxide
radical (NO
2
•) and peroxynitrite (ONOO
−
). Under normal physiological condi-
tions, generation and elimination of these radicals are maintained under redox
balance.
Redox homeostasis is mediated via Nrf2 and NF-
κ
B translocation [25], and
enzymatic and non-enzymatic antioxidant defenses [26]. Enzymes of the anti-
oxidant system that protect against ROS are superoxide dismutase, catalase and
glutathione peroxidase (
Figure 1). One of the main antioxidant enzymes is su-
peroxide dismutase (SOD) which is a metallo-enzyme found in the mitochon-
dria and cytosol that catalyze the conversion of oxygen radicals to hydrogen pe-
roxide, H
2
O
2
at a high reaction rate [26] [27]. The hydrogen peroxide produced
as a result of SOD activity is detoxified by catalases and glutathione peroxidases.
Catalases (CAT) are heme-containing enzymes that catalyze the breakdown of
H
2
O
2
into water and divalent oxygen. CAT activity is mainly localized in perox-
isomes and in a lesser extent in the cytoplasm of erythrocytes, in the nucleus and
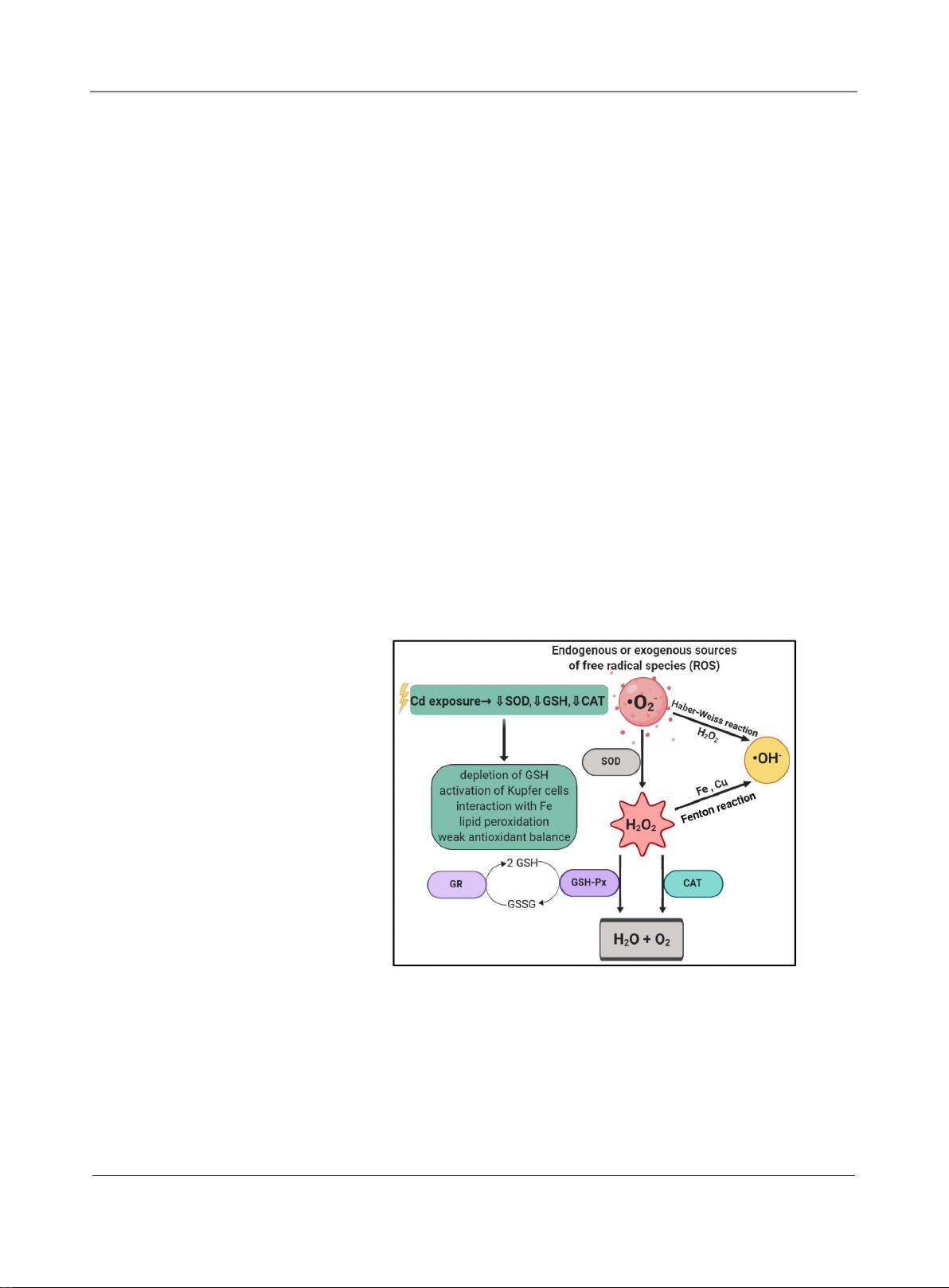
Figure 1. Illustration showing the reactions to maintain redox balance in the cells. Free
radicals like superoxide anions generated by endogenous or exogenous sources is con-
verted to hydrogen peroxide by superoxide dismutase (SOD). The hydrogen peroxide is
further broken down to water and molecular oxygen by either catalase (CAT) or gluta-
thione peroxidase (GSH-Px). Glutathione reductase (GR) and GSH-Px work together to
maintain a balance in glutathione (GSH) and its reduced product (GSSG). The presence
of cadmium in cells interferes with these reactions by decreasing SOD,GSH and CAT
thereby producing OH radicals by Haber-Weiss and Fenton reactions and accumulating
free radicals. Hence, inducing oxidative stress. (Created using biorender.com).

S. C. Das, H. A. Al-Naemi
DOI:
10.4236/odem.2019.74012 147
Occupational Diseases and Environmental Medicine
in the mitochondria. While CAT is active in severe stress conditions, peroxidases
(Px) are suggested to protect the cell during acute oxidative stress. Unlike CAT,
Px detoxify H
2
O
2
through the oxidation of other organic substrates. Glutathione
peroxidases (GSH-Px) utilizes H
2
O
2
to oxidize GSH to glutathione disulfide
(GSSG). Another enzyme involved in the antioxidant defense system is gluta-
thione reductase (GR) which reduces GSSG to GSH. Both GSH-Px and GR work
together in the cells to maintain a steady state in the ratio of GSH to GSSG [28].
When the cells are under oxidative stress, the enzymes of the antioxidant defense
system respond by altering their activity to overcome the oxidative stimuli.
When the concentration of ROS exceeds the threshold such that it is not con-
trolled by antioxidants like ascorbic acid and glutathione or radical scavenging
enzymes like catalase, peroxidase and superoxide dismutase, oxidative damage
to various biomolecules (proteins, lipids and DNA) may ensue consequently
leading to cytotoxicity and genotoxicity [29]. Apart from the enzyme index, a
key measure of oxidative stress is lipid peroxidation, as indicated by the concen-
trations of malondialdehyde (MDA), which may accumulate as a result of cell
damage [30].
In response to Cd exposure, depending on the route of exposure various cel-
lular defense mechanisms are activated to overcome the toxic effects of Cd.
Among the main defense mechanisms are the induction of metallothionein
(MT) primarily in the kidney and liver, elevation in cellular glutathione and ac-
tivation of antioxidant transcription factor Nrf2. These mechanisms are not
“mutually exclusive” but may work together in a cohesive way to resist
Cd-induced oxidative stress. Following exposure, Cd enters the blood stream via
erythrocytes or albumin and undergoes hepatic conjugation where it complexes
with MT to form Cd-MT which can be filtered out by the glomerulus, reab-
sorbed at the proximal tubule and distal tubule. After its entry into the tubular
cell, lysosomes break the Cd-MT complex to free Cd. This free Cd initiates
damage to the kidneys consequently inducing oxidative stress. Apart from this
free Cd, ROS can also be produced when Fenton metals (like iron and copper)
are displaced by Cd from MT or through the depletion of GSH [27]. Highly ab-
undant in cells, GSH is a target of free Cd ions. Cd-induced depletion of the re-
duced GSH pool leads to the disruption of the redox balance leading to an oxid-
ative environment. Hence, Cd-induced oxidative damage is considered to be a
major mechanism rendering its toxicity to various organs by weakening the an-
tioxidant mechanism.
Cadmium does not induce ROS production directly however, this xenobiotic
metal could generate free radicals indirectly [27] [31]. The ensuing response to
the oxidative stress is dependent on the duration of exposure – acute or chronic.
In the case of acute Cd exposure, free radicals inclusive of superoxide anion, hy-
drogen peroxide, hydroxy radical and lipid radicals are generated. These
Cd-induced generation of free radicals are mediated by indirect mechanisms
such as depletion of glutathione, activation of Kupffer cells, inflammation and
involvement of iron in the Fenton reactions [24] [32] [33]. However, in chronic
剩余19页未读,继续阅读
资源评论

weixin_38581308
- 粉丝: 2
- 资源: 893
上传资源 快速赚钱
 我的内容管理
展开
我的内容管理
展开
 我的资源
快来上传第一个资源
我的资源
快来上传第一个资源
 我的收益 登录查看自己的收益
我的收益 登录查看自己的收益 我的积分
登录查看自己的积分
我的积分
登录查看自己的积分
 我的C币
登录后查看C币余额
我的C币
登录后查看C币余额
 我的收藏
我的收藏  我的下载
我的下载  下载帮助
下载帮助

 前往需求广场,查看用户热搜
前往需求广场,查看用户热搜最新资源
- 操作简单的Mongodb网页web管理工具,基于Spring Boot2.0支持mongodb集群.zip
- tms-mongodb-web,提供访问mongodb数据的REST API和可灵活扩展的mongodb web 客户端.zip
- SpringBoot整合mongodb学习MongoTemplate和MongoRepository两种方式CRUD使用.zip
- SpringBoot整合MongoDB实现对数据库的CRUD小demo.zip
- Python操作MongoDB数据库的基本一些操作 .zip
- NOSQL数据库监控工具,目前实现了对Redis、MongoDB的监控功能 .zip
- mongoDB数据库的增删改查,以及所需要的配置.zip
- mongodb数据库idea测试.zip
- koa 分别 连接 mysql、mongodb数据库操作.zip
- 基于pytorch实现的人体部件分割源码+模型.zip
资源上传下载、课程学习等过程中有任何疑问或建议,欢迎提出宝贵意见哦~我们会及时处理!
点击此处反馈



安全验证
文档复制为VIP权益,开通VIP直接复制
 信息提交成功
信息提交成功